INTRODUCTORY NOTES
Date of Information
In this annual information form (“Annual Information Form”), BioHarvest Sciences Inc., together with its subsidiaries, as the context requires, is referred to as the “Company”. All information contained in this Annual Information Form is as at December 31, 2024, unless otherwise stated, being the date of the most recently completed financial year-end of the Company, and the use of the present tense and of the words “is”, “are”, “current”, “currently”, “presently”, “now” and similar expressions in this Annual Information Form is to be construed as referring to information given as of that date.
Cautionary Statement Regarding Forward-Looking Statements and Information
This Annual Information Form contains forward-looking statements and information about the Company which reflect management’s expectations regarding the Company’s future growth, results of operations, operational and financial performance and business prospects and opportunities. In addition, the Company may make or approve certain statements or information in future filings with securities regulatory authorities, in news releases, or in oral or written presentations by representatives of the Company that are not statements of historical fact and may also constitute forward-looking statements or forward-looking information. All statements and information, other than statements or information of historical fact, made by the Company that address activities, events or developments that the Company expects or anticipates will or may occur in the future are forward-looking statements and information, including, but not limited to statements and information preceded by, followed by, or that include words such as “may”, “would”, “could”, “will”, “likely”, “expect”, “anticipate”, “believe”, “intends”, “plan”, “forecast”, “budget”, “schedule”, “project”, “estimate”, “outlook”, or the negative or grammatical variations of those words or other similar or comparable words.
Forward-looking statements and information involve significant risks, assumptions, uncertainties and other factors that may cause actual future performance, achievement or other realities to differ materiality from those expressed or implied in any forward-looking statements or information and, accordingly, should not be read as guarantees of future performance, achievement or realities. Although the forward-looking statements and information contained in this Annual Information Form reflect management’s current beliefs based upon information currently available to management and based upon what management believes to be reasonable assumptions, the Company cannot be certain that actual results will be consistent with these forward-looking statements and information. A number of risks and factors could cause actual results, performance, or achievements to differ materially from the results expressed or implied in the forward-looking statements and information. Such risks and factors include, but are not limited to, the following:
·the Company’s negative operating cash flow and the Company’s ability to continue as a going concern;
·the potential political, economic and military instability in Israel, where the Company’s principal place of business, members of the management team, facilities and employees are located;
·the Company’s additional requirements for capital;
·general business risk and liability;
·the Company’s reliance on key business inputs;
·the Company’s reliance on third-party suppliers and white-label manufacturers;
·the Company’s maintenance obligations and facility disruptions
·the success of the Company’s quality control systems;
·the effectiveness and efficiency of the Company’s advertising and promotional expenditures;
·the failure of the Company’s technology to accommodate increased traffic;
·the increasing consumer acceptance of the Internet as a medium of commerce;
·the development and maintenance of the Internet infrastructure;
·a significant downturn in the Company’s subscriptions may not be immediately reflected in its operating results;
·payment-related risks;
·changing consumer preferences;
·risks related to the price of the Company’s products and contract development and manufacturing operation (“CDMO”) services;
·fluctuations in foreign currency exchange rates;
·the recall of the Company’s products as a result of them not having the intended effects or causing undesirable side effects;
·product liability claims in relation to the Company’s products;
·risks related to insurance;
·risks related to the Company’s management of growth;
·computer system failures, cyber-attacks or deficiencies in cyber security;
·conflicts of interest risk;
·risks related to governmental regulations;
·compliance with manufacturing regulations;
·the Company may face manufacturing stoppages and other challenges associated with audits or inspections by regulatory authorities;
·risks associated with enrolling candidates in clinical trials or studies of others for the Company’s nutraceutical and cosmeceutical products;
·delay in achieving or failure to achieve publicly announced milestones by the Company;
·competition risk;
·obsolescence;
·the loss of key personnel by the Company;
·negative results from clinical or other types of scientific studies and adverse safety events;
·risks related to potential intellectual property claims and patent infringement;
·the protection and enforcement of the intellectual property of the Company;
·the expiration and loss of patents;
·third-party license risk;
·the disclosure of proprietary information and trade secrets of the Company to third parties;
·risks related to smaller companies;
·the Company’s operations may be negatively affected by global financial conditions;
·the Company is a “foreign private issuer” and may lose its foreign private issuer status in the future, which could result in significant additional costs and expenses;
·any return on investment from the Common Shares (as defined herein) is not guaranteed;
·the Company may not pay dividends;
·future sales or issuances of debt or equity securities could decrease the value of any existing Common Shares, dilute investors’ voting power, reduce the Company’s earnings per share and make future sales of the Company’s equity securities more difficult;
·holders of Common Shares are at risk for a substantial loss of capital;
·the Company will have broad discretion over the use of the net proceeds from its financings, and it may not use these proceeds in a manner desired by its shareholders;
·the Common Share price has experienced volatility and may be subject to fluctuation in the future based on market conditions;
·market disruption risks could have a material adverse effect on the market price of the Common Shares;
·there is currently no market through which the Company’s securities, other than the Common Shares, may be sold; and
·there is no assurance of a sufficient liquid trading market for the Common Shares in the future.
For further details, see the “Risk Factors” section of this Annual Information Form.
Although the Company has attempted to identify important risks and factors that could cause actual actions, events or results to differ materially from those described in forward-looking statements or information, there may be other factors and risks that cause actions, events or results not to be as anticipated, estimated or intended. Further, any forward-looking statements and information contained herein are made as of the date of this Annual Information Form and, other than as required by applicable securities laws, the Company assumes no obligation to update or revise them to reflect new events or circumstances. New factors emerge from time to time, and it is not possible for management to predict all of such factors and to assess in advance the impact of each such factor on the Company’s business or the extent to which any factor, or combination of factors, may cause actual realities to differ materially from those contained in any forward-looking statement or information. Accordingly, readers should not place undue reliance on forward-looking statements and information contained in this Annual Information Form and the documents incorporated by reference herein. All forward-looking statements and information disclosed in this Annual Information Form are qualified by this cautionary statement.
2
Presentation of Information
The financial statements referred to herein are reported in United States dollars. Unless otherwise indicated, all references in this Annual Information Form to ‘$” or “US$” are to United States dollars, C$ are to Canadian dollars and “NIS” are to new Israeli shekels.
The Company completed a consolidation (the “Consolidation”) of the Common Shares on the basis of thirty-five (35) pre-Consolidation Common Shares for every one (1) post-Consolidation Common Share on June 3, 2024.
Unless otherwise indicated, all information in this Annual Information Form is presented on a post-Consolidation basis.
CORPORATE STRUCTURE
Name, Address and Jurisdiction
The Company is a biotechnology company that has developed a patented botanical synthesis platform technology (the “Botanical Synthesis Platform Technology”), which enables the Company to grow, in bioreactors at an industrial scale, the active and beneficial ingredients in certain fruits and plants without the need to grow the plant itself.
BioHarvest Sciences Inc. is a corporation incorporated under the Business Corporations Act (British Columbia) (‘BCBCA”) and reporting under the securities laws of British Columbia and Ontario and as a “foreign private issuer” under the securities laws of the United States. The common shares in the capital of the Company (the “Common Shares”) are registered under Section 12(b) of the United States Securities Exchange Act of 1934, as amended (the “Exchange Act”).
The Common Shares are listed for trading on the Global Market (the “Nasdaq GM”) of the Nasdaq Stock Exchange, LLC (“Nasdaq”) under the symbol “BHST”, the Frankfurt Stock Exchange under the symbol “8MV”, the Munich Stock Exchange under the symbol “8MV”, the Stuttgart Stock Exchange under the symbol “CA09076J1084.SG” and the Tradegate Exchange under the symbol “8MV”.
The address of the Company's head office is 1140-625 Howe Street, Vancouver, British Columbia V6C 2T6. The registered and records offices of the Company are located at 704-595 Howe Street, Vancouver, British Columbia V6C 2T5.
Intercorporate Relationships
The Company has the following three (3) subsidiaries, BioHarvest Israel Ltd. (“BioHarvest Israel”), BioHarvest Inc. (Delaware) (“BioHarvest Delaware”) and Superfood Nutraceuticals Inc., as set out in the diagram below:
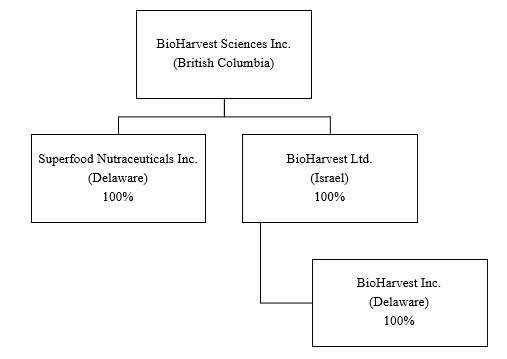
3
GENERAL DEVELOPMENT OF THE BUSINESS
Three-Year History
Over the past three years, the Company has strategically expanded its portfolio of products and services, including the development and launch of VINIA® Superfood Coffee and VINIA® Superfood Tea, and the establishment of the CDMO Services Business Unit.
See the “Business - General” and “Business – Products and Services” sections of this Annual Information Form for additional information on the products and services of the Company and their development during the last three (3) completed financial years.
The following is a summary of the material events during the Company’s last three (3) completed financial years that have influenced the general development of the Company’s business.
Financial Year Ended December 31, 2022
Liron Carmel resigned as a director of the Company on January 10, 2022.
Brian Cornblatt was appointed as the Chief Medical Officer of the Company on May 31, 2022.
The commissioning of the Company’s 20-ton per year manufacturing facility (the “Yavne No. 1 Manufacturing Facility”) in the Yavne No. 1 Property (as defined herein) in Yavne, Israel was completed in Q3 2022.
Financial Year Ended December 31, 2023
The Company decided to discontinue any activity related to the development of medicinal cannabis in February 2023.
Ilana Belzer was appointed as the Chief Operating Officer of the Company on April 19, 2023.
The Company commenced clinical activity in Q2 2023 to assess the ability of the Company’s red grape cell (“RGC”) active ingredient to support additional health and wellness opportunities.
On July 18, 2023, the Company issued C$4,642,075.50 in convertible notes of the Company (“Convertible Notes”) pursuant to a non-brokered private placement. Please see “Market for Securities – Prior Sales” for additional information.
The Company created the CDMO Services Business Unit in Q3 2023.
On October 30, 2023, the Company issued C$11,786,489.98 in Convertible Notes pursuant to a non-brokered private placement. Please see “Market for Securities – Prior Sales” for additional information.
On December 22, 2023, the Company issued C$13,622,873.26 in Convertible Notes pursuant to a non-brokered private placement. Please see “Market for Securities – Prior Sales” for additional information.
In Q4 2023, the Company launched a “functional” VINIA® coffee pod product line, VINIA® Superfood Coffee, consisting of medium roast and decaf coffee pods in the United States (with shipment available in Canada).
Financial Year Ended December 31, 2024
Matthew Zrebiec was appointed as the Vice-President, Business Development, of the Company on March 14, 2024.
Anna Tenstam was appointed as the Vice-President, Business Development, Cosmeceuticals and Injectables, of the Company on March 18, 2024.
On March 20, 2024, Bar Dichter was appointed as the Vice-President, Finance, of the Company, Malkit Azachi was appointed as the Vice-President, Research and Development, of the Company, Michal Sapir was appointed as the Vice-President, Regulatory Affairs, of the Company, and Jared Turner was appointed as the Vice-President, D2C Products Business Unit, of the Company.
4
On March 28, 2024, the Company issued an aggregate of 1,136,444 common share purchase warrants ("Warrants") in consideration for the early conversion of Convertible Notes in the aggregate principal amount and accrued interest of C$16,742,436.10 into approximately 2,093,392 Common Shares, of which 987,515 Warrants are exercisable at an exercise of price of US$7.77 to acquire one (1) Common Share until October 30, 2025 and 148,930 Warrants are exercisable at an exercise price of US$7.77 to acquire one (1) Common Share until December 22, 2025.
On April 18, 2024, the Company issued an aggregate of 88,294 common share purchase warrants (“Warrants”) in consideration for the early conversion of Convertible Notes in the aggregate principal amount and accrued interest of C$5,675,059.91 into approximately 652,175 Common Shares, of which 83,371 Warrants are exercisable at an exercise of price of US$7.77 to acquire one (1) Common Share until October 30, 2025 and 4,923 Warrants are exercisable at an exercise price of US$7.77 to acquire one (1) Common Share until December 22, 2025.
Anne Binder was appointed as a director of the Company on May 27, 2024.
The Company completed the Consolidation of the Common Shares on the basis of thirty-five (35) pre-Consolidation Common Shares for every one (1) post-Consolidation Common Share on June 3, 2024.
On June 21, 2024, the Company issued an aggregate of 114,236 Warrants in consideration for the early conversion of Convertible Notes in the aggregate principal amount and accrued interest of C$1,331,254.50 into approximately 161,620 Common Shares, of which 107,617 Warrants are exercisable at an exercise of price of US$7.77 to acquire one (1) Common Share until October 30, 2025 and 6,619 Warrants are exercisable at an exercise price of US$7.77 to acquire one (1) Common Share until December 22, 2025.
On June 28, 2024, the Company completed a non-brokered private placement of 603,904 Units at a price of US$7.17 per Unit for aggregate gross proceeds of US$4,329,991.68, with each Unit comprising one (1) Common Share, one-quarter of one (1/4) Warrant, with each Warrant exercisable at an exercise price of US$7.68 to acquire one (1) Common Share until December 28, 2024, and one-quarter of one (1/4) Warrant, with each Warrant exercisable at an exercise price of US$11.52 to acquire one (1) Common Share until December 28, 2025.
On June 28, 2024, the Company issued an aggregate of 20,242 Warrants in consideration for the early conversion of Convertible Notes in the aggregate principal amount and accrued interest of C$288,933.09 into approximately 33,697 Common Shares, which Warrants are exercisable at an exercise of price of US$7.77 to acquire one (1) Common Share until December 22, 2025.
In Q2 2024, the Company commenced the building of a new research and development facility (the “Yavne No. 2 R&D Facility”) in the Yavne No. 2 Property (as defined herein) Yavne, Israel to support the CDMO Services Business Unit, which is expected to be completed in Q2 2025.
David Ryan resigned as a director of the Company on September 19, 2024.
Bar Dichter was appointed as the Chief Financial Officer of the Company and ceased to act as the Vice-President, Finance, of the Company, on October 28, 2024, following Alan Rootenberg’s resignation as the Chief Financial Officer of the Company on October 28, 2024.
Matthew Zrebiec resigned as the Vice-President, Business Development, of the Company on November 1, 2024.
The Common Shares ceased trading on the OTCQB on November 11, 2024. The Common Shares were listed and began trading on the Nasdaq GM under the symbol “BHST” on November 12, 2024.
5
The lease for the current location of the Company’s principal place of business in Rehovot, Israel, is likely to be terminated by BioHarvest Israel effective on or before June 2026. The Company intends to move all of its research and development and corporate administrative offices to the Yavne No. 2 Property by Q1 2026.
In Q4 2024, the Company commenced the building of a new manufacturing facility (“Yavne No. 2 Manufacturing Facility”), capable of supporting at least 50 tons per year in the Yavne No. 2 Property in Yavne Israel, to support the demand for VINIA® products and the introduction of new products, such as olive cells and pomegranate cells. The building of the Yavne No. 2 Manufacturing Facility is expected to be completed and start production by Q4 2026.
In Q4 2024, the Company launched a “functional” VINIA® tea product line, VINIA® Superfood Tea, in the United States (with shipment available to Canada).
Subsequent Events to Financial Year Ended December 31, 2024
Sharon Malka was appointed as a director of the Company on January 16, 2025.
Anna Tenstam resigned as the Vice-President, Business Development, Cosmeceuticals and Injectables, of the Company on February 1, 2025.
The Common Shares delisted from and ceased trading on the Canadian Securities Exchange (the “CSE”) on February 14, 2025.
Brian Cornblatt resigned as the Chief Medical Officer of the Company on March 1, 2025.
See “Business – Products and Services” for additional information on changes to the Company’s business expected to occur during the current financial year.
Significant Acquisitions
The Company did not complete any “significant acquisitions” during its most recently completed financial year that required the filing of a “business acquisition report” or other information pursuant to Part 8 of National Instrument 51-102 Continuous Disclosure Obligations.
BUSINESS
General
The Company is a biotechnology company that has developed the Botanical Synthesis Platform Technology, which enables the Company to grow, in bioreactors at an industrial scale, the active and beneficial ingredients in certain fruits and plants without the need to grow the plant itself. The Botanical Synthesis Platform Technology is a non-genetically modified organism platform that can produce plant cells with higher concentrations of active ingredients (as compared to those that are produced naturally)2, as well as high levels of solubility3 and bio-availability4. The
2 The level of resveratrol in VINIA® fresh cells is 845mg/kg (see source (a) below), whereas the average level of resveratrol in fresh grapes grown in a vineyard is 0.69*-3.8* mg/kg (see sources (b) and (c) below):
(a)M. Azachi, R. Yatuv, A. Katz, Y. Hagay, A. Danon, “A novel red grape cells complex: health effects and bioavailability of natural resveratrol.” Int J Food Sci Nutr. 2014 Nov; 65(7):848-55 <https://pubmed.ncbi.nlm.nih.gov/24827888/>.
(b)E. Cantos, J.C. Espín, F.A. Tomás-Barberán, “Varietal differences among the polyphenol profiles of seven table grape cultivars studied by LC-DAD-MS-MS.” J Agric Food Chem. 2002 Sep 25;50(20):5691-6 <https://pubmed.ncbi.nlm.nih.gov/12236700/>.
(c)L. Bavaresco, S. Vezzulli, S. Civardi, M. Gatti, P. Battilani, A. Pietri, F. Ferrari, “Effect of lime-induced leaf chlorosis on ochratoxin A, trans-resveratrol, and epsilon-viniferin production in grapevine (Vitis vinifera L.) berries infected by Aspergillus carbonarius.” J Agric Food Chem. 2008 Mar 26;56(6):2085-9 <https://pubmed.ncbi.nlm.nih.gov/18290620/>.
3 See the study titled “Bioavailability of natural resveratrol from VINIA®” in “Business – Products and Services - Products Business Unit – (a) Nutraceuticals - Scientific Studies” for additional information.
4 The solubility of piceid resveratrol is 25 times greater in water than that of regular trans-resveratrol. The solubility of regular tran-resveratrol is 0.03 g/L (see source (a) below), whereas the solubility of piceid resveratrol/polydatin is 0.766g/L (see source (b) below).
(a)National Library of Medicine, “Compound Summary – Resveratrol - 3.2.4 Solubility” <https://pubchem.ncbi.nlm.nih.gov/compound/445154#section=Solubility> (accessed July 22, 2024).
(b)DrugBank, “Polydatin” <https://go.drugbank.com/drugs/DB11263> (accessed July 22, 2024).
6
Botanical Synthesis Platform Technology is economical, ensures consistency and avoids the negative environmental impacts associated with traditional agriculture by providing consistent product production, a year-round production cycle and products that are devoid of sugar, calories and contaminants, such as pesticides, heavy metals and residues.
The Company owns or has an exclusive license to use twenty-six (26) patents and/or patent applications, of which nineteen (19) patents and/or patent applications cover the Botanical Synthesis Platform Technology and seven (7) patent applications are related to various usages or combinations. Seventeen (17) are granted registered patents. See “Business – Patents” for further details.
The Company is currently focused on utilizing the Botanical Synthesis Platform Technology to develop the next generation of science-based and clinically proven health solutions through its two (2) business units:
1.The Products Business Unit, comprising:
(a)Nutraceuticals: Research, development, manufacturing, marketing and sales of science-based health and wellness nutraceutical solutions which are manufactured and sold as dietary supplements and/or functional food (capsules, powders, chews and other delivery mechanisms such as coffee, teas and electrolyte-enhanced beverages); and
(b)Cosmeceuticals: Research and development for future manufacturing, marketing and sales of science-based health and cosmeceutical solutions which are to be developed, manufactured and sold as cosmetic products; and
2.The CDMO Services Business Unit, a CDMO that offers customers from the pharmaceuticals, cosmeceuticals, nutraceuticals and nutrition industries, through an end-to-end service agreement, the development and manufacturing of specific plant-based active molecules.
As at December 31, 2023, the Company’s business was comprised of two (2) business segments, nutraceuticals and pharmaceuticals. Subsequent to the financial year ended December 31, 2023 and during the three-month period ended March 31, 2024, the Company restructured its operations, such that its operations comprise two (2) business units, the Products Business Unit and the CDMO Services Business Unit, and (i) the nutraceuticals business segment continued into the Products Business Unit and (ii) as for the pharmaceuticals business segment, the Company now offers customers in the pharmaceutical industries the development and manufacturing of specific plant-based active molecules as part of the CDMO Services Business Unit.
The Company previously conducted research into medicinal cannabis and hemp-based products. In February 2023, the Company decided to discontinue any activity related to the development of medicinal cannabis.
Products and Services
As at the date of this Annual Information Form, the Company’s business is comprised of two (2) business units, the Products Business Unit and the CDMO Services Business Unit.
Products Business Unit
(a)Nutraceuticals
The Company is engaged in the research and development of science-based health and wellness solutions for the nutraceutical industry. The Company’s first product entry into this market is a polyphenol/anti-oxidant superfruit product called VINIA®, which is a red grape powder that supplies the benefits of red wine consumption but without the sugar, calories and alcohol found in wine.
VINIA® is made of red grape (Vitis vinifera) cells grown in the Company’s proprietary bioreactor facility. VINIA® is a fine, dry pink-purple powder containing a matrix of polyphenols (with a high concentration of piceid resveratrol) in their natural state (as can be found in red wine) that has additive and synergistic benefits. One of the main active ingredients in VINIA® is piceid resveratrol, maintaining the quality and inherent benefits present in nature without any solvent extraction or genetic modification. VINIA® is soluble when integrated with various liquids or cosmetics.
7
Products
VINIA® powder, in capsule form, is currently sold in the United States (with shipment available to Canada) and Israel, and VINIA® Superfood Coffee and VINIA® Superfood Tea are currently sold in the United States (with shipment available to Canada).
The Company has a well-developed innovation pipeline in its nutraceuticals segment. The Company plans to introduce a number of new products under the VINIA® brand as well as additional cell-based products utilizing the Botanical Synthesis Platform Technology, such as olive cells and pomegranate cells. The existing and planned pipeline of VINIA® products is as follows:
·In Q4 2023, the Company launched a “functional” VINIA® coffee pod product line, VINIA® Superfood Coffee, consisting of medium roast and decaf coffee pods in the United States. These coffee pods are compatible with Keurig®5 and certain single-serve brewing systems. The Company expects to launch coffee pods compatible with the Nespresso®6 brewing system in the United States (with shipment available to Canada) and Israel by Q3 2025;
·In Q4 2024, the Company launched a “functional” VINIA® tea product line, VINIA® Superfood Tea, in the United States (with shipment available to Canada);
·The Company expects to launch VINIA® Superfood Chew supplement in the United States (with shipment available to Canada) by Q3 2025; and
·The Company expects to launch VINIA® hydration powder in the United States (with shipment available to Canada) and Israel by the first half of 2026.
The Company completed the required research and development work related to its new proposed olive cell product (the “Olive Cell Product”), which is expected to target liver and joint health, and the Company expects to obtain all regulatory approvals and complete the required scientific studies to bring Olive Cell Product to market in the United States (with shipment available to Canada) and Israel by the end of 2026. See “Business – Products and Services - Products Business Unit – (a) Nutraceuticals - Regulatory” for additional information.
Regulatory
The Company has conducted various clinical trials and other types of scientific studies to test the efficacy of the VINIA® powder and has submitted notifications to the FDA for the use of its structure/function claims as both a food and dietary supplement on its packaging and in communication materials, which is regulated by the FTC, in the United States.
In November 2011, the Company completed a self-determination that VINIA® is “Generally Recognized as Safe” (“GRAS”) for certain uses in food. The FDA is not required to be notified of and has not reviewed this GRAS self-determination. Subsequent to this GRAS self-determination, the Company began marketing VINIA® as a food. In 2021, the Company began marketing VINIA® as a dietary supplement in the United States, premised on the Company’s marketing of VINIA® as a food.
VINIA® was approved by the Israeli Ministry of Health as a novel food in April 2013 and as a dietary supplement in February 2016 in the Israeli Market. In September 2023, BioHarvest Israel received a product license from Health Canada, authorizing the sale of VINIA® capsules as a natural health product in Canada. This product license sets out the approved recommended use or purpose (claims) that BioHarvest Israel may use in the future marketing of VINIA® in Canada. Currently, up to ninety (90) days’ supply of VINIA® capsules, VINIA® Superfood Coffee and VINIA® Superfood Tea are permitted to be imported by Canadians for personal use from the United States through the Company’s United States e-commerce website, VINIA.com.
5 The Company and its business, operations and products are not affiliated with, endorsed or sponsored by Keurig Dr. Pepper Inc. or Keurig Green Mountain, Inc. “Keurig®” and “K-Cup®” are registered trademarks of Keurig Green Mountain, Inc.
6 The Company and its business, operations and products are not affiliated with, endorsed or sponsored by Nestlé S.A. or Nestlé Nespresso S.A. Nespresso®” is a registered trademark of Société des Produits Nestlé S.A.
8
For the United States market, the Company plans to follow the same regulatory path for its Olive Cell Product as it employed for VINIA®. Specifically, the Company plans to conduct a GRAS self-determination, and then market the Olive Cell Product as a food and dietary supplement. The Company also plans to conduct clinical trials and other scientific studies to evaluate the effect of the Olive Cell Product on liver and joint health, so that, if successful, the Company will be able to make use of structure/function claims on its labeling and in communication materials for the Olive Cell Product in accordance with FDA and FTC requirements.
For the Israeli market, the Company plans to apply to the Israeli Ministry of Health for approval of the Olive Cell Product as a novel food, which involves making submissions concerning the manufacturing process and characterization of the final product.
The Yavne No. 1 Manufacturing Facility received a manufacturer license and GMP approval from the Israeli Ministry of Health in November 2021, as well as key ISO certifications in October 2021.
See “Business - Governmental Regulations” for additional information.
Research and Development
The Company has invested over US$80 million, primarily in research and development activities, to support the business. This investment has enabled the Company to develop a disruptive technology platform which mirrors nature and allows it to efficiently produce plant cells that are identical to those originally sourced from the parent plant, ensuring optimal bio-availability and efficacy of the secondary metabolites. The Company has termed this platform technology “Botanical Synthesis”. The Botanical Synthesis Platform Technology is a non-GMO platform that can produce plant cells with higher concentrations of active ingredients, as compared to those that are produced by nature, as well as high levels of solubility and bio-availability. The Botanical Synthesis Platform Technology provides consistent product production, a year-round production cycle and products that are devoid of sugar, calories and contaminants such as pesticides, heavy metals and residues).
Facilities and Capacity
In terms of manufacturing capacity, the Company has established the Yavne No. 1 Manufacturing Facility and commenced implementation of the required technology and process improvements to drive significant cost reduction through economies of scale.
The Company completed the biological technology transfer to the Yavne No. 1 Manufacturing Facility in March 2022 and has commenced actively scaling up its manufacturing of VINIA® red grape cells at the Yavne No. 1 Manufacturing Facility. This enables the Company to better meet the increasing demand for VINIA®, which is driven by the United States market as a result of the Company’s marketing activities. The Company continued to focus significant resources in the second half of 2023 to increase its capacity levels to meet the growing demand for VINIA® in the United States market. The Company has invested heavily in additional bioreactor capacity and new downstream harvesting and drying equipment in the Yavne No. 1 Manufacturing Facility, which will enable a significant yield increase in the finished product and, accordingly, a corresponding reduction in COGS. In addition, the Company has secured additional third-party drying capacity to be able to cope with growing quarter-on-quarter demand levels.
In April 2024, the Company entered into the Yavne No. 2 Lease (as defined herein) for a property comprising approximately 10,300 square meters in Yavne, Israel (the “Yavne No. 2 Property”), which will comprise the Yavne No. 2 R&D Facility to support the CDMO Services Business Unit upon completion and space to support the building of the Yavne No. 2 Manufacturing Facility to expand the Company’s capacity to produce VINIA® and other products upon completion.
Scientific Studies
Below is a summary of the Company’s scientific studies, including clinical studies, which support the structure/function claims for VINIA® as a food product and dietary supplement/nutraceutical.
9
Study | Summary |
Effect of VINIA® (Grape Cell (RGC) Powder) supplementation on blood pressure and vascular function in people with hypertension7 (Clinical study) | Objectives: To evaluate the effect of VINIA® (RGC powder) supplementation on blood pressure and vascular function in people with mild to moderate hypertension. The primary objective was to evaluate the effect of RGC supplementation on blood pressure and vascular function, specifically, flow-mediated dilatation (“FMD”). The secondary objective was to evaluate the effect of RGC supplementation on the change in oxidative stress parameters. Study design: The study was a randomized, double-blind, placebo-controlled study which lasted three (3) months. After signing an informed consent, subjects were screened for eligibility during two (2) screening visits. Eligible subjects were enrolled and randomized into three (3) treatment arms with daily supplementation for three (3) months of 200 mg RGC powder, 400 mg RGC powder or placebo. Subjects attended the Unit of Clinical Nutrition at the Tel Aviv Sourasky Medical Center every 14±3 days for a total of nine (9) visits. Telephone appointments were conducted during the weeks with no scheduled clinic visits. The subjects were requested not to change their diet or the level of their physical activity for the duration of the study. Number of subjects: 60 planned, 50 recruited and analyzed. Diagnosis and main criteria for inclusion: Subjects aged 35-70 with a body mass index of less than 40.0 kg/m2, systolic blood pressure (SBP) of ≤ 154 mmHg, diastolic blood pressure (“DBP”) of ≤ 93 mmHg and no history of chronic disease. Test product, dose and mode of administration: RGC was supplied in a powder form containing 200 mg or 400 mg. The placebo contained 200 mg of maltodextrin powder with the addition of ponceau 4R (E-124) and brilliant blue FCF (E-133) food colorings. The RGC or placebo was ingested once daily ten (10) minutes before breakfast with a glass of water. Duration of treatment: once daily for 90 days (12 weeks). Statistical analysis: All measured variables were listed individually and, if appropriate, tabulated by descriptive statistics. For descriptive statistics, summary tables were provided giving sample size, absolute and relative frequency by study group, and sample size, arithmetic mean, standard deviation, median, minimum and maximum by study group for means of continuous variables. The following statistical tests were used to study the changes in study parameters. After checking for normal distribution, a Paired T-Test or Signed Rank test (as appropriate) was applied to test the statistical significance of the changes from baseline within each study group. The analysis of variance (“ANOVA”) model was applied to test the statistical significance of the difference in the changes between study groups. Student T-test or non-parametric Rank sum test (as was appropriate) was applied to test the statistical significance of the difference in quantitative parameters (changes and relative changes) between the study groups. The chi-square test or Fisher's Exact test (as was appropriate) was applied to test the statistical significance of the difference for categorical variables (% subjects with positive change) between the study groups. All tests applied were two-tailed, and a p value of 5% or less was considered statistically significant. The data was analyzed using the SAS® version 9.1.3 (SAS Institute, Cary, North Carolina). References: SAS/STAT User’s Guide, SAS Institute Inc. and SAS Procedures Guide, SAS Institute Inc. Efficacy: A significant improvement in FMD measurement and the positive change in FMD was found in the RGC 400 mg arm compared to placebo (p=0.0130 and 0.0294, respectively). A significant statistical decrease was observed in DBP following treatment in the group receiving 200 mg RGC compared to the placebo (p=0.032). A significant decrease in lipid peroxidation and the relative change in lipid peroxide levels were found in subjects on the RGC 400 mg arm (p=0.056 and p=0.052, respectively). A significant difference was detected within the combined RGC groups, mean LDL oxidation-TBARS value decreased from 586.9±48.8 nmol/ml at baseline to 556±34.9 nmol/ml after 12 weeks (p=0.013). |
7 N. Vaisman, E. Niv, “Daily consumption of red grape cell powder in a dietary dose improves cardiovascular parameters: a double blind, placebo-controlled, randomized study.” Int J Food Sci Nutr. 2015 May; 66(3):342-9 <https://pubmed.ncbi.nlm.nih.gov/25666417/>.
10
Bioavailability of natural resveratrol from VINIA®8 (Clinical study) | A bioavailability study was performed in which doses of VINIA® equivalent to 50 and 150 mg of resveratrol were given to fifteen (15) healthy volunteers in a double-blind cross-over design. Blood samples were taken at fourteen (14) time points to compose a pharmacokinetic profile of resveratrol. The human pharmacokinetic analysis revealed relatively high bioavailability and two (2) distinctly separated plasma concentration peaks at one (1) and five (5) h, which was different from competitors with synthetic resveratrol or resveratrol from polygonum, which revealed only one (1) peak.9 VINIA® in the tested doses was found to be safe with no serious adverse events. |
The effect of VINIA® on healthy moderately trained cyclists10 (Clinical study) | A randomized placebo-control study was conducted on healthy moderately trained cyclists. 45 subjects received VINIA® at doses of either 200 mg or 1000 mg or placebo for six (6) weeks. Before and after VINIA® consumption, the subjects underwent anthropometric measurements and exercise testing. A significant 5% to 6% decrease in resting diastolic blood pressure was seen only in the two (2) VINIA® groups after supplementation. The similar effect seen in both VINIA® groups suggests this is a true phenomenon related to VINIA®. There was also a lower mean resting systolic blood pressure, with nearly twice the number of participants decreasing it in both VINIA® groups at the end of the study, yet with no statistical significance. |
The effect of VINIA® on Type 2 Diabetes11 (Clinical trial) | A randomized, double-blind placebo-controlled trial was conducted on subjects with type 2 diabetes. 33 subjects received 1000 mg of VINIA® powder or placebo once daily for 12 weeks. At the end of the treatment period subjects were tested for changes in mRNA expression of the clock genes and type 2 diabetes parameters: fasting and post-prandial circulating levels of HbA1c, glucose, insulin, C-peptide and lipids. A statistically significant reduction in mRNA expression of clock genes (BMAL1, PER 2, Cry 1, CLOCK and RevErb) was detected in white blood cells of patients receiving VINIA®, but not the placebo. This reduced expression may reflect a phase shift (probably phase advance) in clock gene expression. A possible phase advance of clock genes accompanied possible health benefits affecting diabetes and cardiovascular health properties. Moreover, the VINIA® group HbA1c was significantly reduced from baseline in the VINIA® group by 0.55% (p=0.0353) and by 0.16% in the control group (p=0.2334). The difference in changes from baseline between the VINIA® group and the control group was p= 0.0563. Moreover, an ANOVA statistical analysis was performed within the subgroup of HbA1c 7.5-10.1. The difference in changes from baseline between the VINIA® and control group was significantly reduced by 0.82% (p=0.0247). In addition, C-peptide was reduced in the VINIA® group and elevated in the control group with a significant difference between the groups (P =0.0409 Wilcoxon test; P =0.0117 t-test). As a result, the estimated insulin sensitivity calculated from fasting glucose and C-peptide was increased by 40.6% with VINIA® versus control (p<0.0137). According to these results, VINIA® may improve insulin resistance state. C-peptide, a part of being a marker of insulin concentrations, is a bioactive peptide with effects on vascular and microvascular blood flow and tissue health. C- peptide is an important factor for cardiovascular disease events and related mortality. C-peptide was reduced in the VINIA® group and elevated in the control group with a significant difference between the groups (P =0.0117 t-test). |
Evaluation of the effect of VINIA® in metabolic syndrome model on rats12 (Preclinical study) | In a preclinical study on the metabolic syndrome model of rats fed with a high fructose diet, VINIA® (200, 400 and 800 mg per kg of food per day) given in food was found to attenuate the increase in blood pressure, plasma triglycerides and insulin in these animals. |
8 M. Azachi, R. Yatuv, A. Katz, Y. Hagay, A. Danon, “A novel red grape cells complex: health effects and bioavailability of natural resveratrol.” Int J Food Sci Nutr. 2014 Nov; 65(7):848-55 <https://pubmed.ncbi.nlm.nih.gov/24827888/>.
9 D.M. Goldberg, J. Yan, G.J. Soleas, “Absorption of three wine-related polyphenols in three different matrices by healthy subjects.” Clin Biochem. 2003 Feb;36(1):79-87 <https://pubmed.ncbi.nlm.nih.gov/12554065/>.
10 Unpublished.
11 J. Wainstein, O. Froy, Z. Landau, Y. Bar-Dayan, M. Boaz, T. Ganz, M. Menaged, Y. Hagay, M. Azachi, D. Jakubowicz, “Effect of Red Grape Cells, a High Resveratrol Polyphenols Complex, on Glycemic Control, HbA1c, and Clock Gene mRNA Expression in Type 2 Diabetes.” Presented at the American Diabetes Association’s 76th Scientific Sessions (2016) <http://app.core-apps.com/tristar_ada16/abstract/e44e3c20f4b9cf62bd1796c12c1c1cd0>.
12 A. Leibowitz, Z. Faltin, A. Perl, Y. Eshdat, Y. Hagay, E. Peleg, E. Grossman, “Red grape berry-cultured cells reduce blood pressure in rats with metabolic-like syndrome.” Eur J Nutr. 2013 Apr; 53(3):973-80 <https://pubmed.ncbi.nlm.nih.gov/24158651/>.
11
Demonstration of the mechanism of action of VINIA®13 (Invitro study) | The incubation of human umbilical vein endothelial cells (HUVECs) with VINIA® demonstrated a concentration-dependent inhibition of endothelin 1 (“ET-1”) secretion and an increase in the level of endothelial nitric oxide synthase (“eNOS”), signaling a positive effect of VINIA® on vasodilatation and anti-inflammatory activity. eNOS, a vasodilator peptide, and ET-1, a vasoconstrictor protein, are important factors in maintaining vascular tone connection to atherosclerosis. |
(b)Cosmeceuticals
Since Q1 2023, the Company has spent significant resources investigating the opportunities that exist for its RGC molecules in the growing beauty and cosmetics skincare market. The skin care market in the United States is worth approximately US$23 billion as of 202314, and the Company believes that consumers are searching for new natural and natural origin molecules to better address their skin care needs.
In Q1 2023, the Company conducted a small-scale skin care assessment in Seoul, South Korea (the “Skin Care Assessment”), which received positive anecdotal feedback from all participants regarding their various skin ailments, such as atopic dermatosis, psoriasis, facial redness and folliculitis, after using VINIA®. After a ten (10) day selection period, each of the twelve (12) selected participants for the Skin Care Assessment took one (1) VINIA® capsule in the morning for the first two (2) weeks and then took two (2) VINIA® capsules per day thereafter for an additional three (3) weeks. At the conclusion of the assessment, all twelve (12) participants reported positive feedback, including noting skin health promotion.
Based on the results of the Skin Care Assessment, the Company intends to conduct further studies to assess the efficacy of VINIA® as both a dietary supplement and a topical cosmetic solution on skin health promotion, with a view to launching a VINIA® topical solution product (the “VINIA® Topical Solution”) in the United States by the end of 2026.
VINIA®, as a dietary supplement, will be required to comply with the FDA’s requirements with respect to structure/function claims on its packaging and in communication materials in the United States, and the VINIA® Topical Solution will be required to comply with the FDA’s requirements with respect to cosmetics. See “Business - Governmental Regulations” for additional information.
CDMO Services Business Unit
In Q1 2024, the Company announced the launch of its CDMO Services Business Unit, including its entry into a development agreement (collectively, the “Initial Development Agreements”) with each of a food product distributor and Nasdaq-listed biopharmaceutical company (collectively, the “Initial Partners”) to develop complex molecules. In Q4 2024, the Company entered into a development agreement (with the Initial Development Agreements, the “Existing Development Agreements”) with Tate & Lyle PLC, a food product and beverage supplier (with the Initial Partners, the “Existing Partners”).
This CDMO Services Business Unit allows pharmaceutical, cosmeceutical, nutraceutical and nutrition industry companies the opportunity to partner with the Company to utilize the Botanical Synthesis Platform Technology through a CDMO contracting model. The Botanical Synthesis Platform Technology enables the development and manufacturing of patentable plant-based small molecules, complex molecules and unique compositions, which include both small and complex molecules. The Botanical Synthesis Platform Technology can develop complex molecules, otherwise known as biologics, which have a number of unique advantages, including lower costs of development and manufacturing, a faster speed of development and non-immunogenic properties that enhance safety. As a result of these advantages, the Company has decided to name these unique plant-derived complex molecules BIOLOGICS+. BIOLOGICS+ will help address unmet needs in the health industry across pharmaceutical, nutraceutical, cosmeceutical and nutrition verticals.
Under each Existing Development Agreement, the Company has been engaged by such Existing Partners to develop a single plant/cell culture line, containing a unique botanical composition that can predict the future
13 A. Leibowitz, Z. Faltin, A. Perl, Y. Eshdat, Y. Hagay, E. Peleg, E. Grossman, “Red grape berry-cultured cells reduce blood pressure in rats with metabolic-like syndrome.” Eur J Nutr. 2013 Apr; 53(3):973-80 <https://pubmed.ncbi.nlm.nih.gov/24158651/>.
14 Statista, “Skin Care – United States” <https://www.statista.com/outlook/cmo/beauty-personal-care/skin-care/united-states> (accessed May 3, 2024).
12
industrial scale values of its components, through the utilization of the Botanical Synthesis Platform Technology. Upon successful completion of the first phase of development, the Company and such Existing Partner may agree to proceed with additional phases of development. Each Existing Development Agreement has a term of up to three (3) years, subject to earlier termination. The aggregate potential fees that the Company can receive in respect of the services provided to the Existing Partners under the Existing Development Agreements is US$475,000, based on the current stage of development for each Existing Development Agreement. As of the date hereof, the Company has received an aggregate of US$282,000 in respect of the services provided to the Existing Partners under the Existing Development Agreements.
While the Existing Development Agreements validate the Company’s creation and continued development of the CDMO Services Business Unit, the Company does not consider them material contracts based on the relatively low revenue generated therefrom in the context of the Company’s overall revenue.
The Company is focusing its resources on signing additional agreements and commencing and completing the development work required, as applicable.
Manufacturing and Supply
The Company uses several proprietary formulas to feed the red grape cells at different stages of their growth cycle to produce the VINIA® powder, and all ingredients in the proprietary formulas are in stable global supply with stable pricing.
The Company has partnered with a third-party white-label manufacturer for the final manufacturing of the VINIA® Superfood Coffee and VINIA® Superfood Tea, which incorporate the VINIA® powder, which is produced by the Company, and 100% Arabica coffee (in the case of VINIA® Superfood Coffee) and green, black and herbal tea (in the case of VINIA® Superfood Tea), both of which are in stable global supply and subject to global market prices.
The Company uses several proprietary formulas to feed plant cells at different stages of their growth cycle in the research and development phases of its CDMO services, and all ingredients in the proprietary formulas are in stable global supply with stable pricing.
Regulatory Environment
Overview
The foods, food ingredients and dietary supplements that the Company develops are regulated under a number of national and state/provincial laws, including, but not limited to, the United States Food, Drug, and Cosmetic Act (“FDC Act”), as administered by the FDA, the Israeli Public Health Regulations (Food) (Prohibitions on Attributing Healing Attributes to Food Products) (the “PHR”), as administered by the Israeli Ministry of Health, and the Canadian Natural Health Product Regulations (the “NHPR”), as administered by Health Canada.
In the United States, the Company is marketing VINIA® products in reliance on a determination supported by experts that VINIA® is “GRAS” for its intended use. This GRAS determination has not been reviewed by the FDA. If the FDA challenges the GRAS status of an ingredient, the Company could be required to undergo the food additive approval process in order to legally market the product. This process is time-consuming and costly and would interrupt the Company’s business. Furthermore, the Company cannot guarantee that, in such a situation, the use of such ingredient would be approved, and the Company’s business, financial condition and results of operations would be adversely affected.
GMP regulations establish requirements governing the methods, equipment, facilities, and controls for the sanitary production of food. Those who manufacture, package, or hold human food must comply with the GMP regulations.
From time to time in the future, the Company may become subject to additional laws or regulations administered by national, state/provincial or local regulatory authorities, such as the FDA, the FTC, the Israeli Ministry of Health and Health Canada.
Generally, the Company also may become subject to the repeal of national or state/provincial laws or regulations that the Company generally considers favorable or to more stringent interpretations of current laws or regulations. The Company is not able to predict the nature of such future laws, regulations, repeals or interpretations, and it cannot predict what effect additional governmental regulation, if and when it occurs, would have on its business in
13
the future. Such developments could, however, require reformulation of certain products to meet new standards, recalls or discontinuance of certain products not able to be reformulated, additional record-keeping requirements, increased documentation of the properties of certain products, additional or different labeling, additional scientific substantiation, additional personnel or other new requirements. Any such developments could have a material adverse effect on the Company’s business.
The marketing and labeling of any food product in recent years have brought increased risk that consumers will bring putative class action lawsuits and that national, state/provincial or local regulatory or governmental authorities will bring legal action concerning the truth and accuracy of the marketing and labeling of the product. Examples of causes of action that may be asserted in a putative consumer class action lawsuit include fraud, unfair trade practices and breach of consumer protection laws and regulations (such as Proposition 65 in California). national, state/provincial or local regulatory or governmental authorities may bring legal action that seeks removal of a product from the marketplace, fines, and penalties. Even when not merited, putative class claims and actions by national, state/provincial or local regulatory or governmental authorities can be expensive to defend and adversely affect the Company’s reputation with existing and potential customers and consumers and the Company’s corporate and brand image.
See “Risk Factors - Governmental regulations” for additional information.
Food Product Regulation
The Company is also subject to regulation under various international, national, state/provincial and local laws that include provisions governing, among other things, the formulation, manufacturing, packaging, labeling, advertising and distribution of food products.
Food products generally are not required to receive regulatory approval prior to introduction into the United States market. However, a comprehensive regulatory framework governs the safety, manufacture (including composition and ingredients), packaging, labeling and advertising of food in the United States. Principally under the authority of the FDC Act, the FDA:
·regulates manufacturing practices for foods through its current good manufacturing practices regulations;
·specifies the standards of identity for certain foods; and
·prescribes the format and content of certain information required to appear on food product labels.
The FDC Act prohibits the distribution and/or sale of misbranded and/or adulterated foods. The FDA has broad authority to enforce the provisions of federal law, including the power to monitor claims made in product labeling, to seize adulterated or misbranded products or unapproved new drugs, to request product recall, to enjoin further manufacture or sale of a product, to issue warning letters and to institute criminal proceedings.
With the exception of color additives, food ingredients can generally be classified as food additives or GRAS substances, based on the conditions of their intended use. In particular, a food additive is a substance, the intended use of which results or may reasonably be expected to result, directly or indirectly, in it becoming a component of food or otherwise affecting the characteristics of food. Food additives require premarket approval. Excluded from the definition of “food additive” are substances that are generally recognized, among qualified experts, as having been adequately shown through scientific procedures to be safe under the conditions of their intended use or GRAS. A company may establish GRAS status through a “self-determination,” whereby the company determines that a substance is GRAS under the conditions of its intended use, with the assistance of a panel of qualified experts as appropriate. The FDA may disagree with a company’s GRAS determination and require the submission of a food additive petition, and may act to prohibit the marketing of products containing the substance pending approval of the petition.
In markets outside the United States, the Company is usually required to obtain approvals, licenses or certifications from a country’s ministry of health or comparable agency. In Israel, the sale of the Company’s products requires the approval of and is regulated by the Israeli Ministry of Health as a novel food for food/food ingredient products and dietary supplement. In Canada, the sale of the Company’s products requires the approval of and is regulated by Health Canada as a natural health product under the NHPR.
The Company must also comply with labeling regulations in respect of its products. In the United States, conventional foods must meet the FDA’s labeling requirements, including the declaration of Nutrition Facts. The labeling of a conventional food may include a claim regarding the effect of the food on a structure or function of the
14
body, provided that the effect derives from the food’s nutritive value. Such claims are not subject to notification to or premarket approval by the FDA. In the United States, the labeling of dietary supplements is regulated by the FDA under the United States Fair Packaging and Labeling Act (the “FPLA”) and the FDC Act. The labeling of a dietary supplement may include different types of claims, including nutrient content claims, health claims, structure/function claims, statements of nutritional support and certain descriptive claims, subject to compliance with the FDA’s regulations in respect of the labeling of dietary supplements. In Canada, the Company’s products, as natural health products, must comply with specific labeling requirements under the NHPR. In Israel, the Company’s products, as a novel food (food/food ingredient products and dietary supplement), must comply with the labeling requirements of the Israeli Standard 1145 (Labeling of Prepacked Food), Israeli dietary supplement regulations (Public Health Regulation (Food) (Dietary Supplement), 1997) and those set in the directives issued by the Israeli Ministry of Health in approving such products of the Company.
Claims for food or dietary supplements may not imply that the food or dietary supplement is intended to treat, cure, mitigate or prevent a disease, as this causes the product to be an unapproved new drug which may not be marketed without pre-market approval by the FDA. The FDA uses the objective intent of a product’s manufacturer or distributor, as evidenced by the manufacturer or distributor’s labeling claims, advertising matter, or oral or written statements, to determine whether the product is an unapproved new drug.
Manufacturers must ensure their food products or dietary supplements are produced in a sanitary environment to prevent adulteration by contamination, in compliance with the GMP regulations. Those regulations include requirements for sanitation of buildings, facilities, personnel, equipment, and utensils. Those regulations also require processes and controls to ensure safety and prevent contamination from any source. Failure to adhere to GMP requirements can result in a finding that products produced in the facility are adulterated, even if there is no evidence that the products are actually contaminated. If the Company, its suppliers or white-label manufacturers fail to comply with the GMP regulations, the FDA, the Israeli Ministry of Health and Health Canada may take enforcement action against the Company, its suppliers or its white-label manufacturers and seek removal of the Company’s products from the market.
Advertising, including claims regarding the effects or benefits of a product, is regulated by the FTC, Ad Standards and the Israeli Ministry of Health, which may review labeling and advertising materials, including online and television advertisements, to determine whether advertising is truthful and not misleading, and adequately substantiated. Enforcement actions may result in consent decrees, cease and desist orders, judicial injunctions, the payment of fines and other outcomes with respect to advertising claims that are found to be unsubstantiated.
The Company believes it is in compliance with all material governmental regulations applicable to its products.
Cosmetic Regulation
The Company plans to market the VINIA® Topical Solution as a cosmetic in the United States. In the United States, the FDA regulates the labeling of cosmetics under the FPLA and the FDCA, which require that certain information appears on cosmetic labels and that cosmetics are not misbranded (for instance, if the labeling is incorrect or its packaging is misleading or deceptive). FDA approval of cosmetic labels is not required prior to the marketing and sale of cosmetics.
Consumer Laws
The Company sells VINIA® products through its e-commerce websites, the Amazon® Marketplace and its call center. In connection with this, the Company will be subject to various national and state/provincial consumer protection laws, including laws protecting the privacy of consumer information and regulations prohibiting unfair and deceptive acts and trade practices. In particular, under national and state/provincial privacy laws and regulations, the Company may be required to provide consumers with:
·notice of the Company’s policies on sharing non-public information with third parties;
·advance notice of any changes to its policies; and
·with limited exceptions, the right to prevent sharing of their non-public personal information with unaffiliated third parties.
Furthermore, the growth and demand for online commerce could result in more stringent consumer protection laws that impose additional compliance burdens on online retailers. These consumer protection laws could result in substantial compliance costs and could interfere with the conduct of the Company’s business.
15
Specialized Skill and Knowledge
The Company relies on employees with specialized skills and knowledge in cell biology, data science and other disciplines, and the Company believes that such specialized skills and knowledge are available to the Company as they are broadly available in the nutraceutical industry and related industries in Israel.
Competitive Conditions
Products Business Unit
The Company faces competition in the nutraceutical market in which the Company currently operates and the cosmeceutical market in which it intends to operate in the future. Some of the Company’s competitors may be better positioned to develop superior products at lower costs and able to better adapt to changing market conditions than the Company. The Company’s ability to compete depends on, among other things, consistently high product quality, short lead-time, timely delivery, competitive pricing, range of product offerings and superior customer service and support. Increased competition in the markets in which the Company operates may force it to reduce its product prices or may result in increased costs and may have a material adverse effect on its business and operating results. Any decrease in the quality of the Company’s products or level of service to customers or any forced decrease in product pricing may adversely affect its business and operating results.
The Company believes that the Botanical Synthesis Platform Technology provides an advantage over its competitors in the nutraceutical and cosmeceutical markets, as it is a platform technology to solve, on an industrial and commercial scale, the challenge of (i) consistently growing plant cells which mirror the phytonutrients contained in the original plant and (ii) significantly amplifying certain phytonutrient levels without genetic engineering, all without growing such plant and with the added result of removing certain negative components.
Until recently, scientists have been limited to small-scale production with product consistency challenges. Through its use of the Botanical Synthesis Platform Technology, the Company’s unique capabilities can solve major technical challenges in achieving equivalency and consistency across large-scale production. The breakthrough is due to the focus on the parent plant cell as the genesis point of the process to convert it into a powdered form of a whole food phytochemical-rich equivalent product.
CDMO Services Business Unit
The Company’s competition in the CDMO market includes a number of full-service contract manufacturers and large pharmaceutical companies offering third-party development and manufacturing services to fill their excess capacity. Large pharmaceutical companies have been seeking to divest portions of their manufacturing capacity, and any such divested businesses may compete with the Company in the future. In addition, most of the Company’s competitors may have substantially greater financial, marketing, technical or other resources than the Company does. Moreover, additional competition may emerge and may, among other things, result in a decrease in the fees paid for the Company’s services, which would affect the Company’s results of operations and financial condition.
While the Company believes that the advantages of the Botanical Synthesis Platform Technology will enable the Company to compete effectively against other technologies and providers of technology for biologic product development and manufacturing, many of the Company’s competitors have significantly greater financial resources and expertise in research and development and manufacturing. Smaller or early-stage companies may also prove to be significant competitors, particularly through arrangements with large and established companies. In addition, these third parties compete with the Company in recruiting and retaining qualified scientific and management personnel.
See “Risk Factors - Competition risk” for additional information.
Cycles and Seasonality
The Company’s business is not affected by seasonality.
16
Components and Economic Dependence
The Company’s business is not substantially dependent on any particular sales or supply contracts. See “Manufacturing and Supply” for additional information.
Patents
VINIA® products are made from in vitro-grown RGCs without genetic manipulation, including products in the form of a powder. RGC powder, for instance, contains a matrix of polyphenols, including piceid resveratrol, quercetin, anthocyanins, tannins, vitamins, proteins and fibers. The RGC powder is a composition comprising a cell culture of RGC grown in vitro in a large scale-up process.
VINIA® is derived from the red grape skin, flesh, pulp and seed. Accordingly, VINIA® is a source of the whole matrix of nutrients and polyphenols found throughout the red grape and in red wine in their natural state without any genetic manipulation and contains polyphenols (resveratrol, tannins, quercetin, catechins, anthocyanins), vitamins, proteins and fibers. VINIA®’s advantages are the absence of solvent residuals and sugar that has a typical taste of red grapes without bitterness or being astringent.
The Company owns or has an exclusive license to use twenty-six (26) patents and/or patent applications, of which nineteen (19) patents and/or patent applications cover the Botanical Synthesis Platform Technology and seven (7) patent applications are related to various usages or combinations, as described below. Seventeen (17) are granted registered patents.
Jurisdiction | Title | Status | Application No. | Filing Date | Registration No. | Registration Date | Expiration Date |
European Patent Office (4 patents: approved in (i) Germany, (ii) France, (iii) Great Britain and (iv) Italy) | Grape Cell Culture for Treating Inflammation | Registered (1) | 6711231.8 | February 23, 2006 | EP1871402 | July 26, 2017 | February 23, 2026 |
Israel | Grape Cell Culture for Treating Inflammation | Registered (1) | 185476 | February 23, 2006 | 185476 | July 1, 2017 | February 23, 2026 |
Japan | Grape Cell Culture for Treating Inflammation | Registered (1) | 2007-556710 | February 23, 2006 | 5432455 | December 13, 2013 | February 23, 2026 |
United States | Methods for Treating Inflammatory Disorders (3) | Registered (1) | 11/884,774 | September 20, 2007 | 8216801 | July 10, 2012 | February 23, 2026 |
United States | Composition of Cultured Grape Cells (4) | Registered (1) | 13/400,173 | February 20, 2012 | 8628965 | January 14, 2014 | February 23, 2026 |
United States | Composition of Cultured Grape Cells (5) | Registered (1) | 14/097,395 | December 5, 2013 | 9061053 | June 23, 2015 | February 23, 2026 |
China | Process for the Large Scale Production of Fruit Cells | Registered (2) | 201380073592.3 | December 24, 2013 | ZL 201380073592.3 | December 11, 2017 | December 24, 2033 |
17
European Patent Office (two (2) patents: approved in (i) Germany and (ii) Great Britain) | Process for the Large Scale Production of Fruit Cells and Treatment of Diseases With Such Cells | Registered (2) | EP13866675.5 | December 24, 2013 | 2938197 | April 24, 2019 | December 24, 2033 |
Israel | Process for the Large Scale Production of Fruit Cells and Treatment of Diseases With Such Cells | Registered (2) | 239646 | December 24, 2013 | 239646 | August 1, 2019 | December 24, 2033 |
Japan | Process for the Large Scale Production of Fruit Cells and Treatment of Diseases With Such Cells | Registered (2) | 2015-550206 | December 24, 2013 | 6371777 | August 8, 2018 | December 24, 2033 |
United States | Process for the Large Scale Production of Fruit Cells and Treatment of Diseases With Such Cells (6) | Registered (2) | 14/655,052 | June 24, 2015 | 9867861 | January 16, 2018 | December 24, 2033 |
Israel | Pomegranate Derived Cell Culture | Registered (2) | 246525 | January 5, 2015 | 246525 | June 26, 2021 | January 5, 2035 |
United States | Pomegranate Derived Cell Culture | Registered (2) | 15/109,649 | July 4, 2016 | 10,538,737 | January 21, 2020 | January 5, 2035 |
Patent Cooperation Treaty | Method of Treating Raynaud’s Conditions or Erectile Dysfunction | Application filed (2) | PCT/IL2024/05037 | February 6, 2024 | - | - | February 6, 2044 |
Patent Cooperation Treaty | Method of Treating Skin Condition or Disease | Application filed (2) | PCT/IL2024/050147 | February 7, 2024 | - | - | February 7, 2044 |
United States | Olive Derived Cell Culture | Application pending (2) | 18/628,751 | April 7, 2024 | - | - | January 27, 2036 |
Patent Cooperation Treaty | Coffee composition | Application filed (2) | PCT/IL2024/051121 | November 27, 2024 | - | - | November 27, 2044 |
United States | Olive Derived Cell Culture | Provisional (2) | 63/707,284 | October 15, 2024 | - | - | October 15, 2025 |
United States | Tea composition | Provisional (2) | 63/721,588 | November 18, 2024 | - | - | November 18, 2025 |
18
United States | Treatment of colorectal cancer | Provisional (2) | 63/728,201 | December 5, 2024 | - | - | December 5, 2025 |
United States | Combination of OCP/RCP for liver diseases | Provisional (2) | 63/739,534 | December 28, 2024 | - | - | December 28, 2025 |
United States | Treatment of peripheral neuropathy | Provisional (2) | 63/759,502 | February 17, 2025 | - | - | February 17, 2026 |
Notes:
(1)Owned by State of Israel, Ministry of Agriculture & Rural Development, Agricultural Research Organization, The Volcani Center and licensed to BioHarvest Israel pursuant to the License Agreement (as defined herein). See “Material Contracts - License Agreement” for further details.
(2)Owned by BioHarvest Israel.
Changes to Contracts
The Company’s business is not expected to be materially affected in the current financial year by the renegotiation or termination of any contracts or sub-contracts. See “Manufacturing and Supply” for additional information.
Employees
As at December 31, 2024, the Company had 87 employees.
Foreign Operations
The Company’s principal markets for its VINIA® products and CDMO services are Israel and the United States.
The Company’s total revenue in Israel and the United States for the financial year ended December 31, 2024 were US$2,177,000 and US$23,011,000, respectively.
The Company is subject to foreign currency risk. See “Risk Factors – Fluctuations in foreign currency exchange rates” for additional information.
Bankruptcy and Similar Procedures
The Company has not had any bankruptcy, receivership or similar proceedings or any voluntary bankruptcy, receivership or similar proceedings within the three most recently completed financial years and does not anticipate having any such proceedings, and no such proceedings are proposed, during the current financial year.
Reorganizations
Other than as disclosed below, there are no material reorganizations of the Company within the three most recently completed financial years or proposed for the current financial year.
Social or Environmental Policies
The Company is committed to carrying out all of its activities in an ethical manner that prioritizes health and safety, recognizes the concerns of indigenous peoples, communities, local stakeholders and preserves the natural environment.
The Company ensures that all employees are trained and instructed in their assigned tasks and that safety procedures are followed at times. The importance of ethical behavior and preservation of the natural environment is stressed to all employees and contractors, and all are charged with monitoring operations to ensure they are being carried out in an environmentally-friendly manner.
19
RISK FACTORS
There are numerous and varied risks, known and unknown, that may prevent the Company from achieving its goals. The risks described below are not the only ones the Company will face. If any of these risks actually occurs, the Company business, financial condition or results of operations may be materially and adversely affected. In that case, the trading price of the Company’s securities could decline and investors in such securities could lose all or part of their investment.
Risks Relating to the Company
Negative operating cash flow and going concern.
The Company has negative cash flow from operating activities and has historically incurred net losses, including for the financial year ended December 31, 2024. As of December 31, 2024, the Company has an accumulated deficit of US$96,418,000. The Company generated negative cash flows from operating activities of US$6,684,000 and a loss in the amount of US$12,913,000 for the financial year ended December 31, 2024.
There is no assurance that the Company will generate sufficient revenues from sales of its current and future products and the provision of contract development and manufacturing operation (“CDMO”) services in the near future or at all. The Company expects to need to raise additional funds through issuances of securities or through loan financing. There is no assurance that additional capital or other types of financing will be available if needed or that these financings will be on terms acceptable to the Company, or otherwise at least as favorable to the Company as those previously obtained, or at all. Importantly, the inclusion in note 1B to the Company’s audited annual consolidated financial statements for the financial year ended December 31, 2024 and in “Substantial Doubt about the Company’s Ability to Continue as a Going Concern” in the report thereto of Ziv Haft Certified Public Accountants (Isr.) (PCAOB ID No. 1185), a BDO member firm, of disclosure regarding the Company’s ability to continue as a going concern may negatively impact the Company’s ability to raise future financing. If the Company is unable to obtain additional financing from outside sources and eventually generate enough revenues to sustain its operations, the Company may be forced to sell a portion or all of the Company’s assets or curtail or discontinue the Company’s operations. If any of these events happen, investors may lose all or part of their investment.
Potential political, economic and military instability in Israel, where the principal place of business, members of the management team, facilities and employees are located.
The Company’s principal place of business and its facilities, where most of its employees are employed, are located in Rehovot and Yavne, Israel. In addition, the majority of the Company’s key employees and senior management are Israeli citizens. Accordingly, political, economic, and military conditions in Israel may directly affect the Company’s business. Since the establishment of the State of Israel in 1948, a number of armed conflicts have taken place between Israel and groups in its neighboring countries, Hamas (an Islamist militia and political group that has controlled the Gaza Strip) and Hezbollah (an Islamist militia and political group based in Lebanon). While Israel has entered into peace agreements with Egypt, Jordan, the United Arab Emirates, Bahrain, Morocco and Sudan, it has no peace arrangements with any other neighboring or other Arab countries. In addition, relations between Israel and Iran continue to be hostile due to the fact that Iran is perceived by Israel as a sponsor of Hamas and Hezbollah, maintains a military presence in Syria and is viewed as a strategic threat to Israel in light of its nuclear program. The assassinations of Iran’s senior generals Qassim Soleimani by the United States military and Mohsen Farichasde, which Iran claims is associated with Israel, have contributed to the tension in the region and further intensified the hostility between Iran and Israel and between Israel and Hezbollah, which is positioned alongside Israel’s northern border. In addition, several countries, principally in the Middle East, restrict doing business with Israel, and additional countries may impose restrictions on doing business with Israel and Israeli companies, whether as a result of hostilities in the region or otherwise. The restrictive laws, policies, or practices directed towards Israel or Israeli businesses could, individually or in the aggregate, have a material adverse effect on the Company’s business in the future, for example, by way of sales opportunities that the Company could not pursue or from which the Company will be precluded. In addition, should the movement for boycotting, divesting, and sanctioning Israel and Israeli institutions (including universities) and products become increasingly influential in the United States, Canada and elsewhere, this may also adversely affect the Company’s business and financial condition. Further deterioration of Israel’s relations with the Palestinian Authority or countries in the Middle East could expand the disruption of international trading activities in Israel, may materially and negatively affect the Company’s business conditions, could harm the Company’s results of operations, and adversely affect the market price of the Common Shares.
20
Any hostilities involving Israel, terrorist activities, political instability or violence in the region, or the interruption or curtailment of trade or transport between Israel and its trading partners could adversely affect the Company’s operations and results of operations and the market price of the Common Shares.
The Company’s commercial insurance does not cover losses that may occur as a result of an event associated with the security situation in the Middle East. Although the Israeli government is currently committed to covering the reinstatement value of direct damages that are caused by terrorist attacks or acts of war, the Company cannot assure investors that this government coverage will be maintained or, if maintained, will be sufficient to compensate the Company fully for damages incurred. Any losses or damages incurred by the Company could have a material adverse effect on the Company’s business, financial condition and results of operations.
Further, many Israeli citizens are obligated to perform several days, and in some cases, more, of annual military reserve duty each year until they reach the age of 40 (or older for certain reservists) and, in the event of a military conflict, may be called to active duty. In response to increases in terrorist activity, there have been periods of significant call-ups of military reservists. For example, on October 7, 2023, Hamas terrorists invaded southern Israel and launched thousands of rockets in a widespread terrorist attack on Israel. On the same day, the Israeli government declared that the country was at war, and the Israeli military began to call up reservists for active duty, including a few of the Company’s employees. The Company’s operations could be disrupted by these and future reserve duty call-ups. While none of the Company’s facilities or infrastructure have been damaged since the war broke out on October 7, 2023, the import and export of goods may experience disruptions in and out of Israel as a result of such military conflict. Since the war broke out on October 7, 2023, there have been additional active hostilities, including with Hezbollah in Lebanon, with the Houthi movement which controls parts of Yemen, with Iran and in Syria. During November 2024, a ceasefire in Lebanon was declared. During January 2025, Israel and Hamas agreed to a Gaza three-phase ceasefire agreement and partial hostage release (the “Gaza Ceasefire”), the first six-week phase of such ceasefire began on January 19, 2025. On March 18, 2025, Israel announced a resumption of military operations against Hamas, effectively ending the Gaza Ceasefire. It is possible that these hostilities will escalate in the future into a greater regional conflict, and that additional terrorist organizations and countries will actively join the hostilities.
Prolonged hostilities could result in further military reserve duty call-ups in the future, as well as irregularities in the Company’s supply chain, the movement of components and raw materials into Israel and the Company’s finished products exported from Israel. Such disruption could materially adversely affect the Company’s business, prospects, financial condition and results of operations.
Additional requirements for capital.
Substantial additional financing may be required if the Company is to be successful with the development of its business. No assurances can be given that the Company will be able to raise the additional capital that it may require for its anticipated future program of launching new products, such as cosmeceuticals or new nutraceutical cell-product lines, and expanding existing product lines, such as the VINIA® line of products, and any upgrades or improvements that the Company may need to make to its facilities to support the manufacture of its products or the provision of CDMO services. Any additional equity financing may be dilutive to investors, and debt financing, if available, may involve restrictions on financing and operating activities. There is no assurance that additional financing will be available on terms acceptable to the Company, if at all. If the Company is unable to obtain additional financing as needed, it may be required to reduce the scope of its operations or anticipated expansion or may not be able to further develop its business at all.
General business risk and liability.
Given the nature of the Company’s business, it may, from time to time, be subject to claims or complaints from investors or others in the normal course of business. The legal risk facing the Company and its directors, officers and employees in this respect includes potential liability for violations of securities laws, breach of fiduciary duty or misuse of investors’ funds. Some violations of securities laws and breaches of fiduciary duty could result in civil liability, fines, sanctions or the suspension or revocation of the Company’s right to carry on its existing business. The Company may incur significant costs in connection with such potential liabilities.
Product development.
If the Company cannot successfully develop, manufacture and distribute its products, or if the Company experiences difficulties in the development process, such as capacity constraints, quality control problems or other disruptions,
21
the Company may not be able to develop market-ready commercial products at acceptable costs, which would adversely affect its ability to effectively enter or maintain its position in the market. A failure by the Company to achieve a low-cost structure through economies of scale or improvements in manufacturing processes would have a material adverse effect on its commercialization plans and its business, prospects, results of operations and financial condition.
Reliance on key business inputs.
The Company’s business is dependent on a number of key inputs and their related costs, including raw materials and supplies related to its growing operations, as well as electricity, water and other utilities. Any significant interruption or negative change in the availability or economics of the supply chain for key inputs (e.g. rising energy costs) could materially impact the business, financial condition and operating results of the Company. Any ability to secure required supplies and services or to do so on appropriate terms could also have a materially adverse opinion impact on the business, financial condition and operating results of the Company.
Reliance on third-party suppliers and white-label manufacturers.
The Company intends to maintain a full supply chain for the production of its products. The loss of or disruption to its existing or future suppliers and white-label manufacturers would have a material adverse effect on its business and operational results. With respect to raw ingredient and packaging suppliers, given that the Company imports certain inputs to its products from foreign countries, the Company may be subject to disruptions in supply, time delays, and/or cost increases as a result of economic, political, social or other macro factors occurring in those countries, or globally, which are beyond its control. Such disruptions to the supply of key product inputs would have negative downstream impacts on the Company’s overall product development timeline and costs.
Maintenance obligations and facility disruptions.
The Company’s manufacturing processes and those of any of its white-label manufacturers are vulnerable to operational problems that could impair the ability of their respective facilities to manufacture products and the Company to provide CDMO services. The Company and any of its white-label manufacturers could experience a breakdown in any of its machines or other important equipment, and from time to time, planned or unplanned maintenance outages that cannot be performed safely or efficiently during normal hours of operation. Such disruptions could cause a loss of production of the Company’s products or delay in the provision of CDMO services, which could potentially have a material adverse effect on the business, financial conditions and operating results of the Company.
Success of quality control systems.
The quality and safety of the Company’s products are critical to the success of its business and operations. As such, it is imperative that the Company’s quality control systems and those of its service providers operate effectively and successfully. Quality control systems can be negatively impacted by the design of the quality control systems, the quality of training programs and the degree of adherence by personnel to quality control guidelines.
Effectiveness and efficiency of advertising and promotional expenditures.
The Company’s future growth and profitability will depend on the effectiveness and efficiency of its advertising and promotional expenditures, including its ability to (i) create awareness of its current and future products and CDMO services; (ii) determine the appropriate creative message and media mix for future advertising expenditures; (iii) differentiate its products and CDMO services from those of its competitors and (iv) effectively manage advertising and promotional costs in order to maintain acceptable operating margins. There can be no assurance that advertising and promotional expenditures will result in revenues in the future or will generate awareness of the Company’s current and future products and CDMO services. In addition, no assurance can be given that the Company will be able to manage its advertising and promotional expenditures on a cost-effective basis.
Failure to develop the Company’s technology to accommodate increased traffic could reduce demand for its products and impair the growth of its business.
The Company periodically enhances and expands its technology and transaction-processing systems, network infrastructure and other technologies to accommodate increases in the volume of traffic on its e-commerce websites. Any inability to add software and hardware or to develop and upgrade existing technology, transaction-
22
processing systems or network infrastructure to manage increased traffic may cause unanticipated system disruptions, slower response times and degradation in the sales of its products, including the speed of order fulfillment. Failure to manage increased traffic could harm the Company’s reputation and significantly reduce demand for its products, which would impair the growth of its business. The Company may be unable to improve and increase the capacity of its network infrastructure sufficiently or anticipate and react to expected increases in the use of its e-commerce websites to handle increased volume.
The Company continues to enhance its e-commerce websites to better support new features and functionality expected by its customers. Failure of these to perform as expected could lead to customer dissatisfaction and loss of business.
Increasing consumer acceptance of the Internet as a medium of commerce is important to the success of the Company’s business strategy and future revenue growth.
The majority of the Company’s products are sold through its e-commerce websites, VINIA.com and VINIA.co.il, and Amazon®15 Marketplace. The failure of the Internet to continue developing into a significant commercial medium would harm the Company’s ability to increase its revenue and execute its business strategy. Rapid growth in the acceptance and use of the Internet as an effective medium of commerce is a recent development. The acceptance and use of the Internet may not continue to develop, and a sufficiently broad base of consumers may not adopt and continue to use the Internet as a medium of commerce. The Company relies on purchasers who have historically used traditional means of commerce to purchase goods or transact business. If the Company is to be successful, these purchasers must accept and use the Internet as a means of purchasing goods and services and exchanging information. The Company cannot predict the rate at which these purchasers will do so.
The growth of the market for the Company’s products depends on the development and maintenance of the Internet infrastructure.
The Company’s business is primarily based on selling its products over the Internet, and the success of its business, therefore, depends on the development and maintenance of a sound Internet infrastructure. This includes the maintenance of a reliable network backbone with the necessary speed, data capacity, and security, as well as the timely development of complementary products such as high-speed modems to provide reliable Internet access and services. The Company’s ability to increase the speed and scope of the sale of its products is limited by and depends upon, the speed and reliability of both the Internet. Consequently, as Internet usage increases, the growth of the market for its products depends upon improvements made to the Internet to alleviate overloading and congestion. In addition, any delays in the adoption of new standards and protocols required to govern increased levels of Internet activity or increased governmental regulation may have a detrimental effect on the Internet infrastructure.
The Company recognizes e-commerce revenue from customers’ subscriptions and, therefore, a significant downturn in its business may not be immediately reflected in its operating results.
The Company recognizes certain e-commerce revenue from its customers’ subscriptions. As a result, a significant portion of the revenue the Company reports in each quarter is generated from subscriptions entered into during previous periods. Consequently, a decline in new or renewed subscriptions in any one quarter may not impact the Company’s financial performance in that quarter but might negatively affect its e-commerce revenue in future quarters. If a number of subscriptions expire and are not renewed in the same quarter, the Company’s revenue may decline significantly in that quarter and subsequent quarters. In addition, the Company may be unable to adjust its fixed costs in response to reduced revenue. Accordingly, the effect of significant declines in sales or renewals and market acceptance of the Company’s products may not be reflected in its short-term results of operations.
Payments-related risks.
The Company accepts payments for its products using a variety of methods, including credit cards, debit cards and payment upon delivery. For existing and future payment options the Company offers to its customers, it currently is subject to and may become subject to additional regulations and compliance requirements (including obligations to implement enhanced authentication processes that could result in significant costs and reduce the ease of use of its payments methods), as well as fraud. For certain payment methods, including credit and debit cards, the Company pays interchange and other fees, which may increase over time and raise its operating costs and lower
15 The Company and its business, operations and products are not affiliated with, endorsed or sponsored by Amazon.com, Inc. or Amazon Technologies, Inc. “Amazon®” is a registered trademark of Amazon Technologies, Inc.
23
profitability. Failure to comply with these rules or requirements, as well as any breach, compromise, or failure to otherwise detect or prevent fraudulent activity involving the Company’s data security systems, could result in it being liable for card issuing banks’ costs, subject to fines and higher transaction fees, and loss of its ability to accept credit and debit card payments from its customers, process electronic funds transfers or facilitate other types of online payments, and its business and operating results could be adversely affected.
Changing consumer preferences.
As a result of rapidly changing consumer preferences, which are difficult to predict and over which the Company has little, if any, control, many nutraceutical and cosmeceutical products attain financial success for a limited period of time. Even if the Company’s current and future products find retail success, there can be no assurance that any of these products will continue to see extended financial success. The Company’s success will be dependent upon its ability to anticipate or quickly adapt to changing consumer preferences and develop new products that satisfy such preferences. Even if the Company is successful in introducing new products or developing its current products, a failure to continue to evolve its products to meet consumer preferences could cause a decline in its products’ popularity that could reduce its revenues and harm its business, operating results and financial condition.
Risks related to prices.
As the market for the Company’s products and CDMO services matures, or as new or existing competitors introduce new products or services that compete with the Company’s products and CDMO services, the Company may experience pricing pressure and be unable to renew its agreements with existing customers or attract new customers at prices that are consistent with its pricing model and operating budget. If this were to occur, it is possible that the Company would have to change its pricing model or reduce its prices, which could harm its revenue, gross margin and operating results.
Fluctuations in foreign currency exchange rates.
The Company is subject to foreign currency risk. The strengthening or weakening of the Canadian or United States dollar or new Israeli shekel versus other currencies will impact the translation of the Company’s net revenues generated in these foreign currencies into Canadian or United States dollars or new Israeli shekels. The Company imports certain ingredients in its products from foreign countries and so may become forced to pay higher rates for ingredients as a result of the weakening of the Canadian or United States dollar, new Israeli shekel or euro.
The Company has not entered into arrangements or agreements or purchased instruments to hedge its exchange rate risks, and there is no assurance that the Company will enter into such arrangements or agreements or purchased instruments in the future and, if so, that it will continue to do so in the future. The availability and effectiveness of any hedging transaction may be limited, and as a result, the Company may not be able to successfully hedge its exchange rate risks in the future.
Product recalls as a result of the products not having the intended effects or causing undesirable side effects.
Product manufacturers and distributors are sometimes required to recall or initiate returns of their products for various reasons, including product defects such as contaminations, unintended harmful side effects or interactions with other products, packaging safety and inadequate or inaccurate labeling disclosure. While the Company believes that all of its products and the combinations of ingredients in them are safe when taken as directed, if any of the Company’s products are recalled, it could incur unexpected expenses relating to the recall and any legal proceedings that might arise in connection with the recall. The Company may lose significant revenue due to loss of sales and may not be able to compensate for or replace that revenue.
In addition, the Company’s products may not have the effect intended if they are not taken in accordance with certain instructions, which include certain dietary restrictions. Furthermore, there can be no assurance that any of the products, even when used as directed, will have the effects intended or will not have harmful side effects in an unforeseen way or on an unforeseen cohort. Furthermore, a highly publicized actual or perceived problem with products that the Company supplies could adversely affect the market’s perception of its products, which may result in a decline in demand for products supplied by the Company, thereby reducing the Company’s revenues and operating results, which could have a material adverse effect on the Company’s business.
24
Product liability claims.
Although the Company believes that it maintains adequate insurance coverage, it may from time to time be subject to claims for damages resulting from defects in products that it supplies. Product liability claims, even if unsuccessful, may result in significant litigation costs to defend the claims as well as other costs incurred to remedy the problem, such as product recalls, which could substantially increase the Company’s expenses. Successful or partially successful product liability claims could result in significant monetary liability and could seriously disrupt the Company’s business, particularly if the Company’s insurance coverage is inadequate or unavailable in respect of any such claims.
Insurance.
The Company’s operations, products and services may result in the Company becoming subject to liability for environmental damage, property damage, personal injury or other hazards. The Company has obtained or may obtain insurance in accordance with industry standards to address certain risks; such insurance has limitations on liability that may not be sufficient to cover the full extent of such liabilities. In addition, such risks may not be insurable in all circumstances, or, in certain circumstances, the Company may elect not to obtain insurance to deal with specific risks due to the high premiums associated with such insurance or other reasons. The payment of such uninsured liabilities would reduce the funds available to the Company. The occurrence of a significant event that the Company is not fully insured against, or the insolvency of the insurer of such event, could have a material adverse effect on the Company’s financial position, results of operations or prospects.
Management of growth.
The inability of the Company to successfully manage its growth could have a material adverse effect on its operating results and cause its results from operations to fluctuate. As part of the Company’s growth strategy, it intends to introduce new and expand existing product lines, expand sales of its products to existing and new customers in new and existing territories and develop the CDMO Services Business Unit. The Company’s expense levels are based, in part, on expected future revenues, and the Company is constrained in its ability to reduce expenses quickly if, for any reason, its sales or revenue levels do not meet expectations for a particular quarter or period. Furthermore, rapid expansion may place a significant strain on the Company’s senior management team and other key personnel, as well as its business processes, operations and other resources. The Company’s ability to manage growth will also depend in part on its ability to continue to enhance its management information systems in a timely fashion, particularly if customer demands change in ways that the Company does not anticipate. Any inability to manage growth could result in delivery or service delays of the Company’s products and CDMO services and cancellation of customer orders or engagements, which could have a material adverse effect on the Company’s business.
Computer system failures, cyber-attacks or deficiencies in the Company’s or related parties’ cyber security could result in a material disruption of its product development programs, compromise sensitive information related to the Company’s business or trigger contractual and legal obligations, any of which could potentially expose it to liability or reputational harm or otherwise adversely affect it business and financial results.
The Company has implemented its security measures designed to protect the information (including but not limited to intellectual property, proprietary business information and personal information) in its possession, custody or control. The Company’s internal computer systems and those of current and future third parties (such as vendors, collaborators or others) on which the Company relies may fail and are vulnerable to breakdown, breach, interruption or damage from computer viruses, computer hackers, malicious code, employee error or malfeasance, theft or misuse, denial-of-service attacks, sophisticated nation-state and nation-state-supported actors, unauthorized access, natural disasters, terrorism, war, telecommunication and electrical failures or other compromise. Despite the Company’s security practices, there is a risk that it may be subject to phishing and other cyberattacks in the future. The risk of a security breach or disruption, particularly through cyber-attacks or cyber intrusion, including by computer hackers, foreign governments and cyber terrorists, has generally increased as the number, intensity and sophistication of attempted attacks and intrusions from around the world have increased.
The Company may not be able to anticipate all types of security threats, and it may not be able to implement preventive measures effective against all such security threats. The techniques used by cybercriminals change frequently, may not be recognized until launched, and can originate from a wide variety of sources, including outside groups such as external service providers, organized crime affiliates, terrorist organizations or hostile foreign
25
governments or agencies. The Company’s information technology and other internal infrastructure systems, including corporate firewalls, servers, leased lines and connection to the Internet, face the risk of systemic failure that could disrupt its operations. If such an event were to occur and cause interruptions in the Company’s operations, it could result in a material disruption of its development programs and business operations. For example, the loss of clinical trial data from completed or future clinical trials could result in delays and significantly increase the costs to recover or reproduce the data. Likewise, the Company may rely on third parties in connection with the manufacture of its existing or future products, the performance of the CDMO services and the conduct of clinical trials, and similar events relating to their computer systems could also have a material adverse effect on its business.
To the extent that any disruption or security breach was to result in a loss of, or damage to, its data or applications, or inappropriate use, disclosure of or access to confidential or proprietary information, the Company could incur liability, its competitive position could be harmed and the further development and commercialization of its product candidates or any future product candidates could be hindered or delayed. If the Company were to experience a significant cybersecurity breach of its information systems or data, the costs associated with the investigation, remediation and potential notification of the breach to counterparties, data subjects, regulators or others could be material. In addition, the Company’s remediation efforts may not be successful. Moreover, if the information technology systems of the Company’s vendors, collaborators or other contractors or consultants become subject to disruptions or security breaches, it may have insufficient recourse against such third parties, and it may have to expend significant resources to mitigate the impact of such an event and to develop and implement protections to prevent future events of this nature from occurring. If the Company does not allocate and effectively manage the resources necessary to build and sustain the proper technology and cybersecurity infrastructure, it could suffer significant business disruption, including transaction errors, supply chain or manufacturing interruptions, processing inefficiencies, data loss or the loss of or damage to intellectual property or other proprietary information. Furthermore, any such event that leads to unauthorized access, use or disclosure of personal information, including personal information regarding clinical trial participants or employees, could harm the Company’s reputation, compel it to comply with federal and/or state/provincial breach notification laws, cause it to breach its contractual obligations, subject it to mandatory corrective action, and otherwise subject it to liability under laws, regulations and contracts that protect the privacy and security of personal information, which could result in significant legal and financial exposure and reputational damages. As cyber threats continue to evolve, the Company may be required to incur significant additional expenses in order to enhance its protective measures or to remediate any information security vulnerability.
The financial exposure from the events referenced above could either not be insured against or not be fully covered through any insurance that the Company maintains. There can be no assurance that the limitations of liability in the Company’s contracts would be enforceable or adequate or would otherwise protect it from liabilities or damages as a result of the events referenced above.
Conflicts of interest risk.
While the consulting and employment agreements between the Company and its directors and officers typically include non-competition clauses, certain of these directors and officers may, from time to time, become involved in other business ventures in the biotechnology, nutraceutical, cosmeceutical, CDMO services and related industries through their direct and indirect participation in corporations, partnerships, joint ventures, and the like that may become potential competitors to the Company. Situations may arise in connection with potential acquisitions or opportunities where the other interests of these directors and officers conflict with or diverge from the Company’s interests. In accordance with the BCBCA, directors who have a material interest in any person who is a party to a material contract or a proposed material contract are required, subject to certain exceptions, to disclose that interest and generally abstain from voting on any resolution to approve the contract. In addition, the directors and officers are required to act honestly and in good faith with a view to the Company’s best interests. However, in conflict-of-interest situations, directors and officers may owe the same duty to another company and will need to balance their competing interests with their duties to the Company. Circumstances (including with respect to future corporate opportunities) may arise that may be resolved in a manner that is unfavorable to the Company.
Governmental regulations.
The processing, manufacturing, packaging, labeling, advertising and distribution of the Company’s products are subject to regulation by one or more national, state/provincial and state regulatory authorities where the Company currently sells and desires to sell its products. Achievement of the Company’s business objectives is contingent, in part, upon compliance with the regulatory requirements, including those imposed by the United States Food and Drug Administration (“FDA”), the Israeli Ministry of Health, Health Canada and other government authorities,
26
enacted by these government authorities and obtaining, where required, all regulatory approvals, for the sale of its products. The Company cannot predict the time required to secure all appropriate regulatory approvals or support claims for its products or the extent of testing and documentation that may be required by government authorities. Any delays in obtaining or failure to obtain or maintain regulatory approvals or compliance with governmental regulations could significantly delay the development of the Company’s products or require the reformulation of the Company’s products, which could have a material adverse effect on the Company’s business, results of operations and financial condition.
Dietary supplements and dietary ingredients that do not comply with regulations may be deemed adulterated or misbranded. Manufacturers and distributors of dietary supplements and dietary ingredients are prohibited from marketing products that are adulterated or misbranded, and regulatory authorities may take enforcement action against any adulterated or misbranded dietary supplement on the market.
If the Company violates applicable regulatory requirements, a regulatory authority may bring enforcement actions against the Company, which could have a material adverse effect on the Company’s business, prospects, financial condition and results of operations. A regulatory authority may not accept the evidence of safety for any new ingredient that the Company may wish to market, may determine that a particular supplement or ingredient presents an unacceptable health risk and may determine that a particular claim or statement of nutritional value that the Company uses to support the marketing of a supplement is an impermissible drug claim, is not substantiated, or is an unauthorized version of a “health claim” or a “structure/function claim” Any of these actions could prevent the Company from marketing particular nutritional supplement products or making certain claims or statements with respect to those products. Regulatory authorities could also require the Company to remove a particular product from the market. Any future recall or removal would result in additional costs to the Company, including lost revenues from any products that the Company is required to remove from the market, any of which could be material. Any product recalls or removals could also lead to an increased risk of litigation and liability, substantial costs and reduced growth prospects.
A regulatory authority in one jurisdiction may interpret claims or products presumptively valid under the laws and regulations of another jurisdiction as illegal under that jurisdiction’s regulations.
Additional or more stringent laws and regulations of dietary supplements and other products have been considered from time to time. These developments could require the reformulation of some products to meet new standards, recalls or discontinuance of some products not able to be reformulated, additional record-keeping requirements, increased documentation of the properties of some products, additional or different labeling, additional scientific substantiation, or other new requirements. Any of these developments could increase the Company’s costs significantly. In addition, regulators’ evolving interpretation of existing laws could have similar effects.
The United States Federal Trade Commission (the “FTC”), the Canadian Ad Standards (“Ad Standards”) and the Israeli Ministry of Health exercise jurisdiction over the advertising of dietary supplements and requires that all advertising to consumers be truthful and non-misleading. These regulatory authorities actively monitor the dietary supplement space and have instituted numerous enforcement actions against dietary supplement companies for failure to have adequate substantiation for claims made in advertising or for the use of false or misleading advertising claims. Failure to comply with applicable regulations could result in substantial monetary penalties, which could have a material adverse effect on the Company’s financial condition or results of operations.
Compliance with manufacturing regulations.
Governmental authorities ensure the quality of food products and dietary supplements/nutraceuticals by carefully monitoring food and dietary supplement manufacturers’ compliance with GMP regulations. The good manufacturing practices (“GMP”) regulations for food products and dietary supplements/nutraceuticals contain minimum requirements for the methods, facilities and controls used in manufacturing, processing and packing of food products and dietary supplements/nutraceuticals. There can be no assurances that the Company’s existing facilities and those of white-label manufacturers will be able to meet the Company’s timetable and requirements. Further, white-label manufacturers must operate in compliance with GMP regulations, and failure to do so could result in, among other things, the disruption of product supplies. The Company’s dependence upon third parties for the manufacture of its products may adversely affect its profit margins and its ability to develop and deliver products on a timely and competitive basis.
27
The Company may face manufacturing stoppages and other challenges associated with audits or inspections by regulatory authorities.
The regulatory authorities may, at any time and from time to time, audit the facilities in which a product of the Company is manufactured. If any such inspection or audit of such facilities identifies a failure to comply with applicable regulations or if a violation of GMP regulations, the product specifications or other applicable regulations occurs independently of such an inspection or audit, the relevant regulatory authority may require remedial measures that may be costly or time-consuming for the Company to implement and that may include the temporary or permanent suspension of commercial sales or the temporary or permanent closure of a facility. Any such remedial measures imposed upon the Company could materially harm its business.
Risks associated with enrolling candidates in clinical trials and other assessments for the Company’s nutraceutical and cosmeceutical products.
There may be significant competition for recruiting candidates in clinical trials and other assessments, and the Company may be unable to enroll the candidates it needs to complete clinical trials and other assessments on a timely basis or at all. The factors that affect its ability to enroll candidates are largely uncontrollable and include, but are not limited to, the following:
·the size and nature of the candidate population;
·the design of the study protocol;
·the competition with other companies for candidates; and
·the perceived risks and benefits of the products under study.
Delays in completing clinical trials and other assessments may prevent the Company from marketing particular nutritional supplement products or making certain claims or statements with respect to those products.
Delay in achieving or failure to achieve publicly announced milestones.
From time to time, the Company may announce the timing of certain events it expects to occur, such as the anticipated timing of results from its research and development of future products, financial achievements and revenue targets. These statements are forward-looking and are based on the best estimates of management at the time relating to the occurrence of such events. However, the actual timing of such events may differ from what has been publicly disclosed. The timing of events such as initiation or completion of a research and development or manufacturing stage of a future product, product lines or CDMO services, the entry into development agreements to provide CDMO services, the entry obtaining regulatory approval or announcement of clinical trials may ultimately vary from what is publicly disclosed. These variations in timing may occur as a result of different events, including the nature of the results obtained during the research and development phase, the timing of the completion of clinical trials or any other event that has the effect of delaying the publicly announced timeline. The Company undertakes no obligation to update or revise any forward-looking information or statements, whether as a result of new information, future events or otherwise, except as otherwise required by law. Any variation in the timing of previously announced milestones could have a material adverse effect on its business plan, financial condition or operating results and the trading price of the Common Shares.
Competition risk.
The Company faces competition in the markets in which the Company operates and intends to operate in the near future. While the Company believes that the Botanical Synthesis Platform Technology provides an advantage over its competitors, some of the Company’s competitors may be or become better positioned to develop superior products and services at lower costs and able to better adapt to changing market conditions than the Company. The Company’s ability to compete depends on, among other things, science-based claims, patentability and protection of intellectual property, consistency, sustainability, high product quality, efficacy, short lead-time, timely delivery, competitive pricing, range of product offerings and superior customer service. Increased competition in the markets in which the Company operates may force it to reduce its product prices or may result in increased costs and may have a material adverse effect on its business and operating results. Any decrease in the quality of the Company’s products or CDMO services or level of service to customers or any forced decrease in product pricing may adversely affect its business and operating results.
The biotechnology, nutraceutical, cosmeceutical and CDMO services industries are intensely competitive and subject to rapid and significant technological change. The Company’s competitors include large, well-established
28
biotechnology, nutraceutical, cosmeceutical and CDMO services companies and academic and research institutions.
Many of the Company’s competitors have substantially greater financial, technical and human resources than the Company has and have significantly greater experience than the Company in scaling up manufacturing operations and obtaining regulatory approvals of products. Accordingly, the Company’s competitors may succeed in obtaining regulatory approval for products more rapidly than it does. The Company’s ability to compete successfully will largely depend on:
·the efficacy and safety profile of the Company’s future products relative to marketed products and other future products in development;
·the Company’s ability to develop and maintain a competitive position in the product and service categories and technologies on which it focuses;
·the time it takes for the Company’s future products to complete clinical development and receive marketing approval;
·the Company’s ability to obtain required regulatory approvals;
·the Company’s ability to commercialize future products and product lines;
·the Company’s ability to establish, maintain and protect intellectual property rights related to the Company’s existing and future products and product lines; and
·the acceptance and promotion of any of the Company’s existing and future products by medical practitioners and social influencers.
Competitors have developed and may develop technologies that could be the basis for products and services that challenge the products the Company is developing and manufacturing or the CDMO services the Company is providing. Some of those products or services may be more effective or less costly than the Company’s products and CDMO services. The success of the Company’s competitors and its products and technologies relative to its technological capabilities and competitiveness could have a material adverse effect on the future development of products, including its ability to obtain the necessary regulatory approvals and the provision of CDMO services. This may further negatively impact the Company’s ability to generate future product development programs. If the Company is not able to compete effectively against its current and future competitors, its business will not grow, and its financial condition and operations will substantially suffer.
Obsolescence.
Maintaining a competitive position requires constant growth, development, strategic marketing, and planning. If the Company is unable to maintain a technological advantage, the Company’s ability to grow its business will be adversely affected, and its products, CDMO services, technologies or operations may become obsolete compared with other products, services, technologies or operations.
Loss of key personnel.
The Company heavily relies on the capabilities and experience of its key executives and scientists, and the loss of any of them could affect its ability to develop its products or provide CDMO services. The loss of the Company’s Chairperson of the board of directors of the Company (the “Board”), Chief Executive Officer or other key members of staff could harm the Company. The Company also depends on its collaborators and advisors, all of whom have outside commitments that may limit their availability to the Company. In addition, the Company’s future success will depend in large part upon its ability to attract and retain highly skilled scientific, managerial manufacturing and regulatory personnel. The Company enters into agreements with scientific and clinical collaborators and advisors, key opinion leaders and academic partners in the ordinary course of its business. The Company cannot predict its success in hiring or retaining the personnel it requires for continued growth. The loss of the services of any of the Company executive officers or other key personnel could potentially harm the Company’s business, operating results or financial condition.
Negative results from clinical trials or studies of others and adverse safety events.
From time to time, studies or clinical trials on various aspects of nutraceutical and cosmeceutical products are conducted by academic researchers, competitors or others. The results of these studies or trials, when published, may have a significant effect on the market for the nutraceutical or cosmeceutical product that is the subject of the study. The publication of negative results of studies or clinical trials or adverse safety events related to the Company’s existing products and future products, or the categories in which the Company’s existing products or
29
future products compete, could adversely affect the Company’s future commercialization efforts, its share price and its ability to finance future development of its future products, and its business and financial results could be materially and adversely affected.
Risks related to potential intellectual property claims and patent infringement.
Biotechnology companies frequently own trademarks and trade secrets and often enter into litigation based on allegations of infringement or other violations of intellectual property rights. While the Company believes that its existing and future products, CDMO services and operations will not violate the intellectual property rights of third parties, the Company may be subject to intellectual property rights claims in the future, and its products may not be able to withstand any third-party claims or rights against their use. Any intellectual property claims, with or without merit, could be time-consuming, expensive to litigate or settle and could divert management resources and attention. An adverse determination could also prevent the Company from offering its products and services to others and may require that it procure substitute products or services for these members.
With respect to any intellectual property rights claim, the Company may have to pay damages or stop using intellectual property found to be in violation of a third party’s rights. The Company may have to seek a license for the intellectual property, which may not be available on reasonable terms and may significantly increase its operating expenses. The technology also may not be available for license to the Company at all. As a result, the Company may also be required to pursue alternative options, which could require significant effort and expense. If the Company cannot license or obtain an alternative for the infringing aspects of its business, it may be forced to limit its product and service offerings and may be unable to compete effectively. Any of these results could harm the Company’s brand and prevent the Company from generating sufficient revenue or achieving profitability.
Protection and enforcement of intellectual property.
The Company’s success will depend in part upon its ability to protect its intellectual property, including licensed intellectual property, and proprietary technologies and upon the nature and scope of the intellectual property protection it receives. The ability to compete effectively and to achieve partnerships will depend on the Company’s ability to develop and maintain proprietary aspects of its technology and to operate without infringing on the proprietary rights of others. The presence of such proprietary rights of others could severely limit the Company’s ability to develop and commercialize its products to conduct existing research and could require financial resources to defend against litigation, which may be in excess of its ability to raise such funds. There is no assurance that the Company’s patents will be sufficient to protect its proprietary technology and gain or keep any competitive advantage that the Company may have or will be upheld in any proceedings brought by any third parties.
The Company relies upon patents, copyrights, trade secrets, unpatented proprietary know-how and continuing technology innovation to protect the intellectual property that it considers important to the development of the Company’s business. The Company relies on various methods to protect its proprietary rights, including patent applications, confidentiality agreements with its partners, collaborators, advisors, consultants, employees, service providers and management that contain terms and conditions prohibiting the unauthorized use and disclosure of its confidential information. However, despite the Company’s efforts to protect its intellectual property rights, unauthorized parties may attempt to copy or replicate its intellectual property. There can be no assurances that the steps taken by the Company to protect its intellectual property will be adequate to prevent misappropriation or independent third-party development of its intellectual property. While the Company believes that competitors are unlikely to have the necessary technical ability, resources or capacity to successfully develop, replicate and/or implement certain of the Company’s technologies, and in particular, the Botanical Synthesis Platform Technology, to the same extent or at all, it may be possible that other companies can duplicate the Company’s technologies and processes.
The patent positions of biotechnology, nutraceutical, cosmeceutical and CDMO services companies can be highly uncertain and involve complex legal, scientific and factual questions for which important legal principles remain unresolved. Patents issued to the Company may be challenged, invalidated or circumvented. To the extent the Company’s intellectual property, including licensed intellectual property, offers inadequate protection or is found to be invalid or unenforceable, the Company is exposed to a greater risk of direct competition. If the Company’s intellectual property does not provide adequate protection against its competitors’ products, its competitive position could be adversely affected, as could its business, financial condition and results of operations. Both the patent application process and the process of managing patent disputes can be time-consuming, expensive and may divert its management’s attention and its resources, and the laws of some foreign countries may not protect the Company’s intellectual property rights to the same extent as the laws of the United States. The Company will be
30
able to protect its intellectual property from unauthorized use by third parties only to the extent that its proprietary technologies, key products and any future products and CDMO services are covered by valid and enforceable intellectual property rights, including patents or are effectively maintained as trade secrets, and provided it has the funds to enforce its rights, if necessary.
Expiration and loss of patents.
While the Company believes that competitors are unlikely to have the necessary technical ability, resources or capacity to successfully develop, replicate and/or implement certain of the Company’s technologies, and in particular, the Botanical Synthesis Platform Technology, to the same extent or at all, the expiration of patents would nonetheless negatively affect the Company’s ability to enforce its rights against competitors. Competitors may potentially reverse engineer the Company’s technologies, including but not limited to the Botanical Synthesis Platform Technology, and compete directly with the Company in any market where a patent is no longer in force. This may require the Company to reduce the sales price of existing products or fees for CDMO services to remain competitive, invest in further research and development to improve products and CDMO service performance and obtain new patent protection or expend additional funds on marketing to promote brand loyalty. The Company may endeavor to extend the patent life through subsequent patent applications. Commercial products will include intellectual property in the form of trade secrets and know-how; however, this alone cannot protect against competitors designing new technologies, products or services directed to the Company’s patented technologies and processes upon expiration of said patents.
Third-party license risk.
The Company may, from time to time, require third-party licenses to effectively develop, manufacture and/or sell its key products or provide the CDMO services, and it may be unable to predict the availability or cost of such licenses.
A substantial number of patents have already been issued to other biotechnology companies. To the extent that valid third-party patent rights cover its products or CDMO services, the Company or its strategic collaborators would be required to seek licenses from the holders of these patents in order to manufacture, use or sell these products and services, and payments under them would reduce its profits from these products and services. The Company is currently unable to predict the extent to which it may wish or be required to acquire rights under such patents, the availability and cost of acquiring such rights, and whether a license to such patents will be available on acceptable terms or at all. There may be patents in foreign countries or patents issued in the future that are unavailable to license on acceptable terms. The Company’s inability to obtain such licenses may hinder or eliminate its ability to manufacture and market its products or provide and market its CDMO services.
Disclosure of proprietary information and trade secrets to third parties.
The Company may, from time to time, rely to a certain extent on third parties in order to develop or manufacture its products or provide CDMO services, and it may share trade secrets with them. The Company seeks to protect its proprietary technology in part by entering into confidentiality agreements and, if applicable, material transfer agreements, collaborative research agreements, consulting agreements or other similar agreements with its collaborators, advisors, employees and consultants prior to beginning research or disclosing proprietary information. These agreements typically restrict the ability of its partners, collaborators, advisors, employees and consultants to publish data potentially relating to the Company’s trade secrets. The Company’s partners and collaborators may have rights to publish data, provided that the Company is notified in advance and may delay publication for a specified time in order to secure intellectual property rights arising from the collaboration. In other cases, publication rights may be controlled exclusively by the Company, although in some cases, the Company may share these rights with other parties. The Company may also conduct joint research and development programs, which may require it to share trade secrets under the terms of research and development collaborations or similar agreements. Despite the Company’s efforts to protect its trade secrets, its competitors may discover its trade secrets, either through breach of these agreements, independent development or publication of information including its trade secrets in cases where the Company does not have proprietary or otherwise protected rights at the time of publication. A competitor’s discovery of the Company’s trade secrets may impair the Company’s competitive position and could have a material adverse effect on its business and financial condition.
Smaller companies.
Market perception of smaller companies may change, potentially affecting the value of investors’ holdings and the ability of the Company to raise further funds through the issue of further Common Shares or otherwise. The share
31
price of publicly traded smaller companies can be highly volatile. The value of the Company’s securities may go down as well as up, and, in particular, the share price may be subject to sudden and large falls in value given the restricted marketability of the Common Shares, results of operations, changes in earnings estimates or changes in general market, economic and political conditions.
The Company’s operations may be negatively affected by global financial conditions.
Global financial conditions continue to be characterized as volatile. In recent years, global markets have been adversely impacted by various credit crises and significant fluctuations in prices, availability and delivery of fuel and energy, metals, and critical components, including as a result of the COVID-19 pandemic and due to significant fluctuations in commodity prices as a result of the ongoing military conflict between Ukraine and Russia and the economic sanctions imposed on Russia in connection therewith. Many industries have been impacted by these market conditions. Global financial conditions remain subject to sudden and rapid destabilizations in response to international events, as government authorities may have limited resources to respond to future crises. A continued or worsened slowdown in the financial markets or other economic conditions, including but not limited to consumer spending, employment rates, business conditions, inflation, fuel and energy costs, consumer debt levels, lack of available credit, the state of the financial markets, interest rates and tax rates, may adversely affect a particular offering of securities of the Company, the Company’s prospects, cash flows, results of operations, investments or financial condition or the value of the Common Shares. Future crises may be precipitated by any number of causes, including natural disasters, geopolitical instability (such as the Russian invasion of Ukraine, the Hamas-Israel war and continuing or escalating conflicts in the Middle East), changes to energy prices or sovereign defaults. If increased levels of volatility continue or in the event of a rapid destabilization of global economic conditions, it may result in a material adverse effect on prices, demand, availability of credit, investor confidence, and general financial market liquidity, all of which may adversely affect a particular offering of securities of the Company, the Company’s prospects, cash flows, results of operations, investments or financial condition or the value of the Common Shares.
The Company is a “foreign private issuer” and, as a result, is not subject to U.S. proxy rules and are subject to the Exchange Act reporting obligations that, to some extent, are more lenient and less frequent than those of a U.S. domestic public company.
The Company reports under the Exchange Act as a non-U.S. company with foreign private issuer status. Because the Company qualifies as a foreign private issuer under the Exchange Act, it is exempt from certain provisions of the Exchange Act that are applicable to U.S. domestic public companies, including (a) the sections of the Exchange Act regulating the solicitation of proxies, consents or authorizations in respect of a security registered under the Exchange Act, (b) the sections of the Exchange Act requiring insiders to file public reports of their share ownership and trading activities and liability for insiders who profit from trades made in a short period of time and (c) the rules under the Exchange Act requiring the filing with the U.S. Securities and Exchange Commission (the “SEC”) of quarterly reports on Form 10-Q containing unaudited financial and other specified information. In addition, foreign private issuers are not required to file their annual report on Form 20-F as promptly as U.S. domestic issuers. In addition, The Company is permitted to disclose limited compensation information for its executive officers on an individual basis. Further, the Company is not required to comply with Regulation FD, which restricts the selective disclosure of material non-public information to, among others, broker-dealers and holders of a company’s securities under circumstances in which it is reasonably foreseeable that the holder will trade in the company’s securities on the basis of the information. These exemptions and leniencies reduce the frequency and scope of information and protections afforded to shareholders of a company that is not a foreign private issuer.
The Company may lose its foreign private issuer status in the future, which could result in significant additional costs and expenses.
The determination of foreign private issuer status is made annually on the last business day of an issuer’s most recently completed second financial quarter, and, accordingly, the next determination will be made with respect to the Company will be on June 30, 2025. In the future, the Company could lose its foreign private issuer status if (a) more than 50% of its outstanding voting securities are owned by U.S. residents and (b) a majority of its directors or executive officers are U.S. citizens or residents, or the Company fail to meet additional requirements necessary to avoid loss of foreign private issuer status. If the Company loses it foreign private issuer status, the Company will be required to file with the SEC periodic reports and registration statements on U.S. domestic issuer forms, which are more detailed and extensive than the forms available to a foreign private issuer. The Company will also have to mandatorily comply with U.S. federal proxy requirements, and its officers, directors and principal shareholders will become subject to the short-swing profit disclosure and recovery provisions of Section 16 of the Exchange Act. As
32
company that is not a foreign private issuer, the Company will incur significant additional legal, accounting and other expenses that it will not incur as a foreign private issuer.
Risks Relating to the Company’s Securities
Any return on investment from the Common Shares is not guaranteed.
There can be no assurance regarding the amount of return to be generated by the Company’s business. The Common Shares are equity securities of the Company and are not fixed-income securities. Unlike fixed-income securities, there is no obligation of the Company to distribute to shareholders of the Company a fixed amount or to return the initial purchase price of a Common Share on a date in the future. The market value of the Common Shares may deteriorate, and that deterioration may be significant.
The Company may not pay dividends.
The Company intends to retain earnings, if any, to finance the growth and development of its business and does not intend to pay cash dividends on the Common Shares in the foreseeable future. The payment of future cash dividends, if any, will be reviewed periodically by Board and will depend upon, among other things, conditions then existing, including earnings, financial condition and capital requirements, restrictions in financing agreements, business opportunities and conditions and other factors.
Future sales or issuances of debt or equity securities could decrease the value of any existing Common Shares, dilute investors’ voting power, reduce the Company’s earnings per share and make future sales of the Company’s equity securities more difficult.
The Company may sell additional equity securities in subsequent offerings (including through the sale of securities convertible into equity securities) and may issue additional equity securities to finance operations, acquisitions or other projects. The Company cannot predict the size of future issuances of equity securities or the size and terms of future issuances of debt instruments or other securities convertible into equity securities or the effect, if any, that future issuances and sales of its securities will have on the market price of the Common Shares. Any transaction involving the issuance of previously authorized but unissued Common Shares, or securities convertible into Common Shares, would result in dilution, possibly substantial, to securityholders. Exercises of presently outstanding share options may also result in dilution to securityholders.
The Board has the authority to authorize certain offers and sales of additional securities without the vote of, or prior notice to, shareholders of the Company. Based on the need for additional capital to fund expected expenditures and growth, it is likely that the Company will issue additional securities to provide such capital. Such additional issuances may involve the issuance of a significant number of Common Shares at prices less than the current market price for the Common Shares.
Sales of substantial amounts of the Company’s securities, or the availability of such securities for sale, could adversely affect the prevailing market prices for the Company’s securities and dilute investors’ earnings per share. A decline in the market prices of the Company’s securities could impair the Company’s ability to raise additional capital through the sale of securities should the Company desire to do so. Sales of the Common Shares by shareholders might also make it more difficult for the Company to sell equity securities at a time and price that the Company deems appropriate.
The Common Share price has experienced volatility and may be subject to fluctuation in the future based on market conditions.
The Common Shares are listed for trading on the Nasdaq GM under the symbol “BHST”, the Frankfurt Stock Exchange under the symbol “8MV”, the Munich Stock Exchange under the symbol “8MV”, the Stuttgart Stock Exchange under the symbol “CA09076J1084.SG” and the Tradegate Exchange under the symbol “8MV”. An investment in the Company’s securities is highly speculative. The market has from time-to-time experienced significant price and volume fluctuations that are unrelated to the financial performance or prospects of any particular company. In addition, because of the nature of the Company’s business, certain factors such as the Company’s announcements, competition, the Company’s financial condition or results of operations as reflected in the Company’s interim and annual financial statements, the Company’s operating performance and the performance of competitors and other similar companies, changes in earnings estimates or recommendations by research analysts who track the Company’s securities or securities of other companies in the Company’s sector,
33
general market conditions, announcements relating to litigation, the arrival or departure of key personnel and the factors listed under “Cautionary Statement Regarding Forward-Looking Statements and Information” and “Risk Factors” and elsewhere in this Annual Information Form can have an adverse impact on the market price of the Common Shares.
Any negative change in the public’s perception of the Company’s prospects could cause the price of the Company’s securities, including the price of the Common Shares, to decrease dramatically. Furthermore, any negative change in the public’s perception of the prospects of biotechnology, nutraceutical, cosmeceutical or CDMO services companies in general could depress the price of the Company’s securities, including the price of the Common Shares, regardless of the Company’s results. In the past, following declines in the market price of a company’s securities, securities class-action litigation often has been instituted against the company. Litigation of this type, if instituted, could result in substantial costs and a diversion of the Company’s management’s attention and resources.
Market disruption risks could have a material adverse effect on the market price of the Common Shares.
War and occupation, terrorism and related geopolitical risks may in the future lead to increased short-term market volatility and may have adverse long-term effects on world economies and markets generally. These risks could also adversely affect securities markets, inflation and other factors relating to the securities that would be held from time to time. Such events could, directly or indirectly, have a material adverse effect on the price of the Common Shares.
Holders of Common Shares are at risk for a substantial loss of capital.
Holders of Common Shares could experience a loss of all or substantially all of their investment in the Company. There can be no assurance that the Company will be able to generate positive returns. Therefore, an investment in the Company should only be considered by persons who can afford a loss of their entire investment.
The Company will have broad discretion over the use of the net proceeds from its financings, and it may not use these proceeds in a manner desired by its shareholders.
The Company will have broad discretion over the use of the net proceeds from its financings. Because of the number and variability of factors that will determine the Company’s use of such proceeds, the Company’s ultimate use might vary substantially from its planned use. Prospective investors may not agree with how the Company allocates or spends the proceeds from its financings. The Company may pursue acquisitions, collaborations or other opportunities that do not result in an increase in the market value of its securities, including the market value of the Common Shares, and that may increase its losses.
There is no assurance of a sufficient liquid trading market for the Common Shares in the future.
Shareholders of the Company may be unable to sell significant quantities of Common Shares into the public trading markets without a significant reduction in the price of the Common Shares or at all. There can be no assurance that there will be sufficient liquidity of the Common Shares on the trading market, and that the Company will continue to meet the listing requirements of the Nasdaq GM or achieve and maintain listing on any other public listing exchange.
There is currently no market through which the Company’s securities, other than the Common Shares, may be sold.
There is currently no market through which the securities of the Company, other than the Common Shares, may be sold and purchasers of securities of the Company, other than Common Shares, may not be able to resell such securities.
Holders of securities, other than Common Shares, have no rights as shareholders of the Company.
Until a holder of securities of the Company, other than Common Shares, acquires Common Shares upon exercise or conversion of such securities, such holder will have no rights with respect to any Common Shares underlying such securities. Upon exercise or conversion of such securities into Common Shares, such holder will be entitled to exercise the rights of a shareholder of the Company only as to matters for which the record date occurs after the exercise or conversion date.
34
DIVIDENDS
To date, the Company has not paid any dividends. The Company intends to retain its earnings, if any, to finance the future growth and development of its business and does not expect to pay dividends or to make any other distributions in the foreseeable future. Payment of dividends in the future is dependent upon the earnings and financial condition of the Company and other factors which the Board may deem appropriate at the time.
There are no restrictions in the constating documents of the Company, and it is not currently expected that there will exist such restrictions elsewhere, which could prevent the Company from paying dividends.
DESCRIPTION OF CAPITAL STRUCTURE
The Company’s authorized capital consists of an unlimited number of Common Shares without par value, of which 17,327,716 Common Shares are issued and outstanding as at the date of this Annual Information Form, and an unlimited number of preferred shares (the “Preferred Shares”) without par value, of which none are issued and outstanding. As of the date of this Annual Information Form, an additional aggregate of 4,029,415 Common Shares have been reserved and allotted for issuance upon the due exercise or vesting of Warrants, stock options (“Options”) and restricted share units (“RSUs”) of the Company.
Common Shares
Holders of Common Shares are entitled to receive notice of any meetings of shareholders of the Company and to attend and cast one (1) vote per Common Share at all shareholder meetings of the Company.
Holders of Common Shares are entitled to receive on a pro-rata basis such dividends on the Common Shares, if any, as and when declared by the Board at its discretion from funds legally available therefor, and upon the liquidation, dissolution or winding up of the Company are entitled to receive on a pro-rata basis the net assets of the Company after payment of debts and other liabilities, in each case subject to the rights, privileges, restrictions and conditions attaching to any other series or class of shares ranking senior in priority to or on a pro-rata basis with the holders of Common Shares with respect to dividends or liquidation. The Common Shares do not carry any pre-emptive, subscription, redemption or conversion rights, nor do they contain any sinking or purchase fund provisions.
Preferred Shares
The Preferred Shares may include one or more series and, subject to the BCBCA, the directors of the Company may, by resolution, if none of the shares of that particular series are issued, alter the Articles and authorize the alteration of the Notice of Articles, as the case may be, to do one or more of the following:
·determine the maximum number of shares of that series that the Company is authorized to issue, determine that there is no such maximum number, or alter any such determination;
·create an identifying name for the shares of that series, or alter any such identifying name; and
·attach special rights or restrictions to the shares of that series, or alter any such special rights or restrictions.
MARKET FOR SECURITIES
Trading Price and Volume
The Common Shares are listed for trading on the Nasdaq GM under the symbol “BHST”, the Frankfurt Stock Exchange under the symbol “8MV”, the Munich Stock Exchange under the symbol “8MV”, the Stuttgart Stock Exchange under the symbol “CA09076J1084.SG” and the Tradegate Exchange under the symbol “8MV”.
The following table sets forth the reported high and low sale prices and the aggregate volume of trading of the Common Shares on the CSE, the Canadian marketplace on which the greatest volume of trading or quotation of the Common Shares occurred during the most recently completed financial year and up until February 14, 2025,
35
for the most recently completed financial year. The Common Shares ceased to trade on the CSE on February 14, 2025.
| High (C$) | Low (C$) | Volume |
January 2024 | 8.05 (1) | 7.35 (1) | 45,045 (1) |
February 2024 | 8.40 (1) | 7.18 (1) | 60,779 (1) |
March 2024 | 10.50 (1) | 7.00 (1) | 56,716 (1) |
April 2024 | 9.80 (1) | 8.05 (1) | 27,984 (1) |
May 2024 | 9.10 (1) | 7.70 (1) | 37,869 (1) |
June 2024 | 9.20 | 7.85 | 37,124 |
July 2024 | 8.50 | 7.99 | 32,652 |
August 2024 | 8.30 | 7.20 | 29,046 |
September 2024 | 7.80 | 7.20 | 49,319 |
October 2024 | 7.60 | 6.24 | 234 |
November 2024 | 8.87 | 6.63 | 686 |
December 2024 | 9.00 | 7.95 | 226 |
Note:
(1)Approximate figures calculated on a post-Consolidation basis.
The following table sets forth the reported high and low sale prices and the aggregate volume of trading of the Common Shares on the Nasdaq GM, the foreign marketplace on which the greatest volume of trading or quotation of the Common Shares currently occurs, for the most recently completed financial year. The Common Shares began trading on the Nasdaq GM on November 12, 2024.
Month and Year | High (US$) | Low (US$) | Volume |
November 12 to 29, 2024 (1) | 6.72 | 5.48 | 217,401 |
December 2024 | 6.85 | 5.91 | 179,496 |
Notes:
(1)The Common Shares began trading on the Nasdaq GM on November 12, 2024.
(2)Approximate figures calculated on a post-Consolidation basis.
Prior Sales
During the most recently completed financial year, the Company issued the following securities not listed or quoted on a marketplace:
Date of Sale or Grant | Security | Number/Principal Amount | Exercise / Deemed Issue Price | Expiry Date |
February 20, 2024 | Options (1) | 13,600 | C$7.875 | February 20, 2026 |
February 20, 2024 | Options (1) | 2,857 | C$7.875 | February 20, 2034 |
March 22, 2024 | Options (1) | 8,571 | C$9.975 | March 22, 2027 |
March 22, 2024 | Options (1) | 14,286 | C$9.975 | June 22, 2027 |
March 22, 2024 | Options (1) | 47,144 | C$9.975 | March 22, 2034 |
March 28, 2024 | Warrants (2) | 987,513 | US$7.77 | October 30, 2025 |
March 28, 2024 | Warrants (2) | 148,931 | US$7.77 | December 22, 2025 |
April 18, 2024 | Warrants (2) | 83,371 | US$7.77 | October 30, 2025 |
April 18, 2024 | Warrants (2) | 4,923 | US$7.77 | December 22, 2025 |
April 26, 2024 | Warrants (2) | 8,571 | C$9.10 | April 26, 2026 |
April 29, 2024 | RSUs (3) | 20,000 | C$9.10 | December 31, 2024 |
May 31, 2024 | Options (1) | 16,429 | C$8.925 | May 31, 2034 |
June 21, 2024 | Warrants (2) | 107,617 | US$7.77 | October 30, 2025 |
June 21, 2024 | Warrants (2) | 6,619 | US$7.77 | December 22, 2025 |
June 28, 2024 | Warrants (2) | 150,978 | US$7.68 | December 28, 2024 |
June 28, 2024 | Warrants (2) | 150,978 | US$11.52 | December 28, 2025 |
June 28, 2024 | Warrants (2) | 20,242 | US$7.77 | December 22, 2025 |
November 15, 2024 | Options (1) | 111,998 | C$8.48 | November 15, 2034 |
November 15, 2024 | RSUs (3) | 5,400 | C$8.48 | November 15, 2025 |
Notes:
(1)Each Option is exercisable at such exercise price to acquire one (1) Common Share until such expiry date, subject to vesting conditions, as set forth in the table above.
36
(2)Each Warrant is exercisable at such exercise price to acquire one (1) Common Share until such expiry date as set forth in the table above.
(3)Each RSU is issued at such deemed issue price until such expiry date, subject to such vesting conditions as set forth in the table above.
ESCROWED SECURITIES
To the knowledge of the management of the Company, no securities of the Company are currently held in escrow or are subject to contractual restrictions on transfer.
DIRECTORS AND EXECUTIVE OFFICERS
The following table sets forth the name, municipality, province or state of residence, position held with the Company, the date of appointment of each director and executive officer, principal occupation within the immediately preceding five years and the shareholdings of each director and executive officer of the Company. The statement as to securities beneficially owned, or controlled or directed, directly or indirectly, by the directors and executive officers named below is in each instance based upon information furnished by the person concerned and is as at the date of this Annual Information Form.
Name and Jurisdiction of Residence | Positions Held and Date of Appointment or Election | Principal Occupation during the past 5 years |
Ilan Sobel Central District, Israel | Chief Executive Officer (June 9, 2020) | Chief Executive Officer of the Company since July 2020; Chief Operating Officer and Chief Commercial Officer of Weissbeerger from November 2014 to June 2020. |
Bar Dichter Central District, Israel | Chief Financial Officer (October 28, 2024) | Chief Financial Officer of the Company since October 2024; Vice-President, Finance, of the Company from March 2024 to October 2024; Vice-President, Finance, of BioHarvest Israel from July 2022 to October 2024; and Controller of BioHarvest Israel from 2018 to July 2022. |
Zaki Rakib Dubai, UAE | President (June 9, 2020) Chairman (June 9, 2020) Director (September 27, 2018) | President and Chairman of the Company since June 2020; Director of the Company since September 2018; Chief Executive Officer of the Company from September 2018 to June 2020; Chief Executive Officer and a director of BioHarvest Israel since September 2018; Director of BioHarvest Delaware since February 2018; and Director of Dolarin Ltd. from July 2014 to December 2021. |
Yoheved (Yochi) Hagay Central District, Israel | Chief Technology Officer (September 27, 2018) | Chief Technology Officer of the Company since September 2018; and Chief Technology Officer of BioHarvest Israel since September 2018. |
Ilana Belzer Central District, Israel | Chief Operating Officer (April 19, 2023) | Chief Operating Officer of the Company since April 2023; and Chief Operating Officer of CollPlant Biotechnologies Ltd. from 2015 to April 2023. |
David Ryan British Columbia, Canada | Secretary (April 19, 2013) Vice-President, Investor Relations (June 9, 2020) | Secretary of the Company since April 2013; Vice-President, Investor Relations, of the Company since June 2020; Director of the Company from April 2013 to September 2024; President of the Company form April 2013 to June 2020; Director of HydroGraph Clean Power Inc. from November 2020 to April 2022; Director of International Battery from August 2019 to March 2022; Director of GlobeX Data Ltd. from March 2017 to May 2020; Chief Executive Officer and director of Scotch Creek Ventures Inc. since January 2017; Director of Ovation Science Inc. from October 2017 to October 2023; Director of InsuraGuest Technologies Inc. since August 2010; and Chief Executive Officer, President and a director of Glenstar Ventures Corp. since April 2013. |
Malkit Azachi Central District, Israel | Vice-President, Research and Development (March 20, 2024) | Vice-President, Research and Development, of the Company since March 2024; Vice-President, Research and Development, of BioHarvest Israel since January 2011. |
37
Michal Sapir Central District, Israel | Vice-President, Regulatory Affairs (March 20, 2024) | Vice-President, Regulatory Affairs, of the Company since March 2024; Vice-President, Regulatory Affairs, of BioHarvest Israel since January 2010. |
Jared Turner Tokyo, Japan | Vice-President, D2C Products Business Unit (March 20, 2024) | Vice-President, D2C Products Business Unit, of the Company since March 2024; Vice-President, E-Commerce and Global Commercial Operations, of BioHarvest Israel since December 2021. |
Vivien Rakib, Tel Aviv District, Israel | Director (September 27, 2018) | Director of the Company since September 2018; Director of BioHarvest Israel since 2007; Director of Shaker since 2011; and Director of CoPro since 2013. |
John (Jake) Fiddick (1)(2)(3) British Columbia, Canada | Director (February 7, 2019) | Director of the Company since February 2019; Director of Makara Mining Corp. from June 2019 to September 2020. |
David Tsur (2) Central District, Israel | Director (October 20, 2021) | Director of the Company since October 2021; Chairman of Kanabo Group Plc from February 2021 to January 2025; and Director of Kamada Ltd. since 1990. |
Anne Binder (1)(2)(3) Île-de-France, France | Director (May 27, 2024) | Director of the Company since May 2024; Director of Lectra Systems SA from 2011 to April 2023; Financial strategy consultant since 1996. |
Sharon Malka (1) Central District, Israel | Director (January 16, 2025) | Director of the Company since January 2025; Chief Executive Officer of Dotz Nano Ltd. since March 2023, Director of MediWound Limited (“MediWound”) from July 2022 to June 2023; Chief Executive Officer of MediWound from May 2019 to June 2022. |
Notes:
(1)Member of the Audit Committee of the Board (the “Audit Committee”), of which Sharon Malka is the Chair.
(2)Member of the Compensation Committee of the Board, of which Anne Binder is the Chair.
(3)Member of the Nominating and Corporate Governance Committee of the Board, of which Anne Binder is the Chair.
Term
Each of the Company’s directors serves until his or her successor is elected at the next annual general meeting of the Company. Each of the Company’s executive officers serves at the pleasure of the Board.
Shareholdings of Directors and Executive Officers
As at the date of this Annual Information Form, the directors and executive officers of the Company, as a group, beneficially owned, controlled or directed, directly or indirectly, 3,338,815 Common Shares, representing approximately 19.27% of the issued and outstanding Common Shares.
Cease Trade Orders or Bankruptcies
To the knowledge of the Company:
(a)no director or executive officer of the Company is, as at the date of this Annual Information Form, or was within 10 years before the date of this Annual Information Form, a director, chief executive officer or chief financial officer of any company (including the Company), that:
(i)was subject to an order that was issued while the director or executive officer was acting in the capacity as director, chief executive officer or chief financial officer; or
(ii)was subject to an order that was issued after the director or executive officer ceased to be a director, chief executive officer or chief financial officer and which resulted from an event that occurred while that person was acting in the capacity as director, chief executive officer or chief financial officer.
For the purposes of this subsection (a), “order” means a cease trade order, an order similar to a cease trade order or an order that denied the relevant company access to any exemption under securities legislation, and in each case that was in effect for a period of more than 30 consecutive days.
38
(b)no director or executive officer of the Company, or a shareholder holding a sufficient number of securities of the Company to affect materially control of the Company:
(i)is, as at the date of this Annual Information Form, or has been within the 10 years before the date of this Annual Information Form, a director, chief executive officer or chief financial officer of any company (including the Company) that, while that person was acting in that capacity, or within a year of that person ceasing to act in that capacity, become bankrupt, made a proposal under any legislation relating to bankruptcy or insolvency or was subject to or instituted any proceedings, arrangement or compromise with creditors or had a receiver, receiver manager or trustee appointed to hold its assets; or
(ii)has, within the 10 years before the date of this Annual Information Form, become bankrupt, made a proposal under any legislation relating to bankruptcy or insolvency, or was subject to or instituted any proceedings, arrangement or compromise with creditors, or had a receiver, receiver manager or trustee appointed to hold the assets of the director, executive officer or shareholder.
The foregoing information, not being within the knowledge of the Company, has been furnished by the respective directors, executive officers and shareholders holding a sufficient number of securities of the Company to affect materially control of the Company.
Penalties or Sanctions
To the knowledge of the Company, no director or executive officer of the Company, or a shareholder holding a sufficient number of securities of the Company to affect materially the control of the Company, has been subject to:
(a)any penalties or sanctions imposed by a court relating to securities legislation or by a securities regulatory authority or has entered into a settlement agreement with a securities regulatory authority; or
(b)any other penalties or sanctions imposed by a court or regulatory body that would likely be considered important to a reasonable investor in making an investment decision regarding the Company.
The foregoing information, not being within the knowledge of the Company, has been furnished by the respective directors, executive officers and shareholders holding a sufficient number of securities of the Company to affect materially control of the Company.
Conflicts of Interest
The Company’s directors and officers may serve as directors or officers of other companies or have significant shareholdings in other companies and, to the extent that such other companies may participate in ventures in which the Company may participate, the directors of the Company may have a conflict of interest in negotiating and concluding terms respecting the extent of such participation. In the event that such conflict of interest arises at a meeting of the Board, a director who has such a conflict will abstain from voting for or against the approval of such participation or such terms. In accordance with the BCBCA, the directors of the Company are required to act honestly, in good faith and in the best interests of the Company. In determining whether or not the Company will participate in a particular program and the interest therein to be acquired by it, the directors will primarily consider the degree of risk to which the Company may be exposed and its financial position at that time.
The directors and officers of the Company are aware of the existence of laws governing the accountability of directors and officers for corporate opportunity and requiring disclosures by the directors and officers of conflicts of interest and the Company will rely upon such laws in respect of any directors’ and officers’ conflicts of interest or in respect of any breaches of duty by any of its directors and officers. All such conflicts will be disclosed by such directors or officers in accordance with the BCBCA and they will govern themselves in respect thereof to the best of their ability in accordance with the obligations imposed upon them by law. See “Risk Factors – Conflicts of Interest Risk”.
As of the date of this Annual Information Form, except as disclosed under “Interests of Management and Others in Material Transactions”, the directors and officers of the Company are not aware of any such existing or potential conflicts of interests.
39
PROMOTERS
During the previous two (2) most recently completed financial years or during the current financial year, no person or company has been a promoter of the Company or any subsidiary of the Company.
AUDIT COMMITTEE
Audit Committee Charter
The text of the Audit Committee Charter, as amended and restated on September 20, 2024, of the Company (the “Audit Committee Charter”) is attached hereto as Schedule “A”.
Composition of the Audit Committee
The Audit Committee consists of Sharon Malka, David Tsur and Anne Binder, of which Mr. Malka is the chair. National Instrument 52-110 Audit Committees (“NI 52-110”) provides that a member of an audit committee is “independent” if the member has no direct or indirect material relationship with the Company, which could, in the view of the Company’s Board, reasonably interfere with the exercise of the member’s independent judgment. All of the Audit Committee members are considered independent. All of the Audit Committee members are “financially literate, as defined in NI 52-110, as all have the industry experience necessary to understand and analyze financial statements of the Company, as well as the understanding of internal controls and procedures necessary for financial reporting.
Relevant Education and Experience of the Audit Committee
All members of the Audit Committee have the ability to read, analyze and understand the complexities surrounding the issuance of financial statements that present a breadth and level of complexity of accounting issues that are generally comparable to the breadth and complexity of the issues that can reasonably be expected to be raised by the Company’s financial statements, and have an understanding of internal controls. The directors of the Company have determined that all members of the Audit Committee are “independent” and “financially literate” for the purposes of applicable laws and the rules of Nasdaq. The directors of the Company have also determined that the Company has two (2) “Audit Committee Financial Experts” for the purposes of applicable laws and the rules of Nasdaq. The designation of a member of the Audit Committee as an “Audit Committee Financial Expert” does not make the member an “expert” for any purpose, impose any duties, obligations or liability on the member that are greater than those imposed on members of the Board who do not carry this designation or affect the duties, obligations or liability of any other member of the Audit Committee.
In addition to each member’s general business experience, the education and experience of each Audit Committee member that is relevant to the performance of his or her responsibilities as an Audit Committee member is as follows:
Sharon Malka, Director
Mr. Malka has held senior leadership positions with a number of international healthcare and technology companies. Since March 2023, he has been the Chief Executive Officer of Dotz Nano Ltd., an Australian-based technology company focused on developing, manufacturing and commercializing advanced materials for diagnostics solutions. Mr. Malka previously served as the Chief Executive Officer (from May 2019 to June 2022), Chief Financial Officer (April 2007 to April 2019) and a director (from July 2022 to June 2023) of MediWound Limited, a Nasdaq-listed biopharmaceutical company, and as a Partner at Variance Economic Consulting Ltd., a financial services consulting boutique focused on international technology companies, from 2002 to 2006
Mr. Malka is a certified CPA (2000) in Israel and a graduate of the Executive Education Program (2021) of Harvard Business School in the United States. He holds a BSc in Business Administration (1998) from The College of Management in Israel and an MBA (2000) from Bar-Ilan University in Israel.
John (Jake) Fiddick, Director
Mr. Fiddick, a director of the Company since February 2019, founded the “Public Company Banking Group” for the Bank of Montreal (“BMO”) in 1984 and was part of a 15-person team specializing in servicing public companies
40
before retiring in August 2017. Mr. Fiddick previously served as a director of Makara Mining Corp. from June 2019 to September 2020.
During his 58 years with BMO, he obtained his real estate license and broker’s license.
Anne Binder, Director
Ms. Binder specializes in the support and financing of small and medium-sized enterprises. Since 1996, she has worked as a financial strategy consultant and independent director for mainly private companies operating in various sectors, such as finance, electronics, integrated technological solutions and luxury. She previously acted as a senior advisor to Tikehau Investment Management SAS, a management company specializing in corporate debt financing. Ms. Binder also previously served as a director of each of Lectra, Oceasoft, CNCEF (Chambre Nationale des Conseils Experts Financiers) and CNCIF (Chambre Nationale des Conseillers en Investissements Financiers) and as a member of the supervisory board of the Financial Investment Agency, Economic Development of Nîmes.
From 1993 to 1996, Ms. Binder was a director in charge of development in France for the international financial services group General Electric Capital, and director of its French subsidiary. From 1990 to 1993, Ms. Binder was the managing director of the holding company and deputy managing director of the EURIS investment fund. From 1983 to 1990, she participated in the creation of the Pallas Group of which she was a director. Ms. Binder was a Vice-President of Générale Occidentale, a bank and industrial holding company, from 1978 to 1982.
Ms. Binder holds a Master in Public Affairs (1971) from Sciences Po in France and an MBA (1974) from INSEAD in France.
Audit Committee Oversight
At no time since the commencement of the Company’s most recently completed financial year was a recommendation of the Audit Committee to nominate or compensate an external auditor not adopted by the Board.
Reliance on Certain Exemptions by the Audit Committee
At no time since the commencement of the Company’s most recently completed financial year has the Company relied on any of the exemptions set out in Section 2.4 (De Minimis Non-audit Services), Section 3.2 (Initial Public Offerings), Section 3.4 (Events Outside Control of Member), Section 3.5 (Death, Disability or Resignation of Audit Committee Member), Subsection 3.3(2) (Controlled Companies), 3.6 (Temporary Exemption for Limited and Exceptional Circumstances) or Section 3.8 (Acquisition of Financial Literacy) of NI 52-110, or an exemption from NI 52-110, in whole or in part, granted under Part 8 of NI 52-110.
Pre-Approval Policies and Procedures of the Audit Committee
The Audit Committee has adopted a specific policy and procedure for the engagement of non-audit services as described in Section 4 of the Audit Committee Charter. The Audit Committee must (i) approve all audit engagement fees and terms (with the power to sign any engagement letter providing for the same on behalf of the Company) and (ii) pre-approve all audit and permitted non-audit and tax services that may be provided by the Company’s independent auditors or other registered public accounting firms, and establish policies and procedures for the Committee’s pre-approval of permitted services by the Company’s independent auditors or other registered public accounting firms on an on-going basis.
External Auditor Service Fees
The aggregate fees billed by the Company’s external auditor in the financial years ended December 31, 2024 and 2023 are disclosed below:
41
Financial Year Ended | Audit Fees (US$) (1) | Audit Related Fees (US$) (2) | Tax Fees (US$) (3) | All Other Fees (US$) (4) | Total |
December 31, 2024 | 315,000 | 36,000 | 4,500 (5) (approximate) | - | 355,500 (approximate) |
December 31, 2023 | 109,000 | - | 4,162 (6) | - | 113,612 |
Notes:
(1)“Audit fees” include fees billed by the Company’s external auditor in each of the last two (2) financial years for audit fees.
(2)“Audit-related fees” include the aggregate fees billed in each of the last two (2) financial years for assurance and related services by the Company’s external auditor that are reasonably related to the performance of the audit or review of the Company’s financial statements and are not reported under “Audit fees” above.
(3)“Tax fees” include the aggregate fees billed in each of the last two (2) financial years for professional services rendered by the Company’s external auditor for tax compliance, tax advice and tax planning.
(4)“All other fees” include the aggregate fees billed in each of the last two (2) financial years for products and services provided by the Company’s external auditor, other than “Audit fees”, “Audit related fees” and “Tax fees” above.
(5)Related to advice on withholding tax in Israel.
(6)Related to services provided in connection with tax filings in the United States.
LEGAL PROCEEDINGS AND REGULATORY ACTIONS
Since the beginning of the Company’s most recently completed financial year, the Company is not aware of: (a) any legal proceedings to which it is a party, or by which any of its property is subject, which would be material to it and are not aware of any such proceedings being contemplated, (b) any penalties or sanctions imposed by a court relating to securities legislation or a securities regulatory authority, or other penalties or sanctions imposed by a court or regulatory body against it that would likely be considered important to a reasonable investor making an investment decision and (c) any settlement agreements that the Company has entered into before a court relating to securities legislation or with a securities regulatory authority.
INTEREST OF MANAGEMENT AND OTHERS IN MATERIAL TRANSACTIONS
To the knowledge of the Company, no director, executive officer or shareholder holding on record or beneficially, directly or indirectly, more than 10% of the issued Common Shares, or any of their respective associates or affiliates has any material interest, direct or indirect, in any transaction in which the Company has participated within the three (3) most recently completed financial years or during the current financial year, which has materially affected or is reasonably expected to materially affect the Company.
The foregoing information, not being within the knowledge of the Company, has been furnished by the respective directors, executive officers and shareholders holding a sufficient number of securities of the Company to affect materially control of the Company.
TRANSFER AGENT AND REGISTRAR
The transfer agent and registrar for the Common Shares is National Securities Administrators Ltd. of Vancouver, British Columbia.
MATERIAL CONTRACTS
Except for contracts entered into in the ordinary course of business and as disclosed below, the Company did not enter into any material contracts in the most recently completed financial year, or before the most recently completed financial year that are still in effect.
42
Yavne No. 1 Agreement
Pursuant to the lease agreement dated January 16, 2025 (the “Yavne No. 1 Agreement”) between BioHarvest Israel and Sugart Ltd. (the “Yavne No. 1 Landlord”), BioHarvest Israel entered into a lease (the “Yavne No. 1 Lease”) for a property, comprising approximately 1,100 square meters, in Yavne, Israel from September 1, 2024 until September 1, 2025, subject to extension for an additional two (2) rental periods of six (6) months each if:
·BioHarvest Israel did not substantially breach the Yavne No. 1 Agreement during the previous rental period, which breach was not corrected within fourteen (14) days of notice by the Yavne No. 1 Landlord; and
·BioHarvest Israel provides 90 days’ notice prior to the end of the previous rental period to extend the Yavne No. 1 Lease.
In consideration for the Yavne No. 1 Lease, BioHarvest Israel pays rent monthly to the Yavne No. 1 Landlord, subject to increase and other adjustments, including based on the Consumer Price Index published by the Israeli Central Bureau of Statistics. As of the date of this Annual Information Form, BioHarvest Israel has paid NIS 706,125 in rent for the Yavne No. 1 Lease.
Equipment Agreement
Pursuant to the agreement dated March 31, 2024 (the “Equipment Agreement”) between BioHarvest Israel and Enlivex Therapeutics R&D, Ltd. (the “Seller”), BioHarvest Israel agreed to assume the Seller’s lease (the “Yavne No. 2 Lease”) to a property comprising approximately 10,300 square meters (the “Yavne No. 2 Property”) in Yavne, Israel, effective April 1, 2024, and to purchase from the Seller certain equipment installed in the Yavne No. 2 Property (the “Equipment”). In consideration for BioHarvest Israel’s purchase of the Equipment, BioHarvest Israel agreed to pay to the Seller an aggregate of NIS 13,000,000 (the “Equipment Purchase Price”) as follows:
·NIS 4,000,000, by April 1, 2024 (which amount has been paid); and
·NIS 9,000,000 (the “Equipment Purchase Price Balance”), in twenty-four (24) equal monthly instalments of NIS 375,000 commencing on April 1, 2024, in respect of which NIS 4,875,000 has been paid as at the date of this Annual Information Form, provided that:
oBioHarvest Israel will be entitled to a 4% discount to the Equipment Purchase Price Balance if the Equipment Purchase Price Balance is prepaid in one (1) installment by October 1, 2025;
oBioHarvest Israel will be entitled to a 2% discount to any portion of the Equipment Purchase Price Balance equal to or greater than NIS 4,000,000 if such portion is prepaid by October 1, 2025;
oIf BioHarvest Israel fails to pay a monthly installment of the Equipment Purchase Price Balance within seven (7) days of the due date for such installment (other than due to a breach by the Seller of the Equipment Agreement), the Seller will be entitled to charge interest at an annual rate of 8% on such installment; and
oIf BioHarvest Israel fails to pay a monthly installment of the Equipment Purchase Price within thirty (30) days of the due date for such installment, BioHarvest Israel and will discuss and attempt to agree on a revised payment schedule within thirty (30) days following the date that is thirty (30) days following the default of such installment payment. If BioHarvest Israel and the Seller fail to agree to a revised payment schedule:
·The Seller will have the right to sell the Equipment and/or assume the Yavne No. 2 Lease (and demand BioHarvest Israel evacuate the Yavne No. 2 Property), subject to providing BioHarvest Israel ninety (90) days’ prior written notice, and further subject to the portion of the Equipment Purchase Price paid by BioHarvest Israel as of the date of such evacuation notice; and
·The Seller will have the right to assume the Yavne No. 2 Lease (and demand BioHarvest Israel evacuate the Yavne No. 2 Property) if BioHarvest Israel fails to make payments to Eitan On Investment Ltd. (the “Yavne No. 2 Landlord”) in accordance with the Yavne No.
43
2 Agreement (as defined herein), subject to providing BioHarvest Israel thirty (30) days’ prior written notice.
The title to the Equipment shall pass to BioHarvest Israel upon payment by BioHarvest to the Seller of the Equipment Purchase Price, and the risk of loss to the Equipment passed to BioHarvest Israel on April 1, 2024.
Tripartite Agreement
In connection with BioHarvest Israel’s entry into the Tripartite Agreement and the Yavne No. 2 Agreement, BioHarvest Israel entered into an agreement dated March 31, 2024 with the Seller and the Yavne No. 2 Landlord, pursuant to which:
·BioHarvest Israel agreed to assume the Yavne No. 2 Lease; and
·The Seller will have the right to re-assume the Yavne No. 2 Lease until April 1, 2027 if BioHarvest fails to pay the Equipment Purchase Price Balance in accordance with the Equipment Purchase Agreement or if the Yavne No. 2 Landlord terminates the Yavne No. 2 Agreement (as defined herein) as a result of a breach by BioHarvest Israel in accordance with the Yavne No. 2 Agreement.
Yavne No. 2 Agreement
Pursuant to the lease agreement dated March 31, 2024 (the “Yavne No. 2 Agreement”) between BioHarvest Israel and the Yavne No. 2 Landlord, BioHarvest Israel entered into the Yavne No. 2 Lease for the Yavne No. 2 Property, comprising approximately 10,300 square meters, from April 1, 2024 until September 30, 2030 (the “Yavne No. 2 Initial Period”), subject to extension for an additional sixty (60) months (the “Yavne No. 2 First Renewal Period”) if:
·BioHarvest Israel did not substantially breach the Yavne No. 2 Agreement during the Yavne No. 2 Initial Period, which breach was not corrected within thirty (30) days of notice by the Yavne No. 2 Landlord; and
·BioHarvest Israel provides ten (10) months’ notice prior to the end of the Yavne No. 2 Initial Period to extend the Yavne No. 2 Lease for the Yavne No. 2 First Renewal Period,
and subject to extension for an additional sixty (60) months (the “Yavne No. 2 Second Renewal Period”) if:
·BioHarvest Israel did not substantially breach the Yavne No. 2 Agreement during the Yavne No. 2 First Renewal Period, which breach was not corrected within thirty (30) days of notice by the Yavne No. 2; and
·BioHarvest Israel provides ten (10) months’ notice prior to the end of the Yavne No. 2 First Renewal Period to extend the Yavne No. 2 Lease for the Yavne No. 2 Second Renewal Period,
In consideration for the Yavne No. 2 Lease, BioHarvest Israel pays rent monthly to the Yavne No. 2 Landlord, subject to an annual increase and other adjustments, including based on the Consumer Price Index published by the Israeli Central Bureau of Statistics. As of the date of this Annual Information Form, BioHarvest Israel has paid NIS 722,794 in rent for the Yavne No. 2 Lease.
License Agreement
Pursuant to the agreement dated March 21, 2007 (the “License Agreement”) between BioHarvest Israel and State of Israel, Ministry of Agriculture & Rural Development, Agricultural Research Organization, The Volcani Center (the “Licensor”), the Licensor granted BioHarvest Israel an exclusive worldwide license, including the right to sub-license, to the rights to six (6) patents (the “Licensed Patents”) as described in “Business – Patents”, in consideration for the payment by BioHarvest Israel to the Licensor of the following payments:
·the equivalent of an aggregate of US$40,000 in NIS (which amount has been paid);
·a royalty fee equal to 3% of all sales (ex-factory) of VINIA® products in countries in which a Licensed Patent has been registered until the expiration of the Licensed Patents on February 23, 2026, subject to renewal (the “Licensed Patent Expiry Date”), in respect of which US$112,091 in NIS has been paid as at the date of this Annual Information Form;
44
·a royalty fee equal to 1.25% of all sales (ex-factory) of VINIA® products in countries in which a Licensed Patent has not been registered until the Licensed Patent Expiry Date, in respect of which no amounts have been payable as at the date of this Annual Information Form; and
·if BioHarvest sub-licenses the rights to the Licensed Patents to a third party, 5% of the consideration received by BioHarvest in respect of such sub-license, in respect of which no amounts have been payable as at the date of this Annual Information Form.
The License Agreement will terminate on the Licensed Patent Expiry Date, subject to the renewal of any Licensed Patents.
TV Network Agreement
Pursuant to the memorandum of agreement dated July 12, 2023 (the “TV Network Agreement”) between BioHarvest Israel and TBN Broadcasting of Texas, Inc. (the “TV Network”), the TV Network will promote the Company through programs, interviews and advertisements and provide BioHarvest with a list of donors to use to offer the Company’s products. In consideration for the TV Network’s services, BioHarvest Israel will pay to the TV Network:
·an amount equal to a percentage between 40% and 50% of the revenue from first orders of the Company’s product(s) by each customer provided by the TV Network; and
·an amount equal to a percentage between 15% and 25% of the revenue from a subsequent order of the same product(s) or any additional orders of other product(s) by each such customer,
and such payments shall be made:
·in cash or Common Shares, at the discretion of the TV Network, such Common Shares to be issued at a price per Common Share equal to the closing price of the Common Shares on the date of the TV Network’s notice (or the trading day prior if no trading took place on such date), prior to the Common Shares being listed on a Nasdaq exchange; or
·in cash, after the Common Shares are listed on a Nasdaq exchange,
in respect of which the TV Network has received US$3,382,607 as at the date of this Annual Information Form.
If, from time to time, BioHarvest Israel obtains:
·25,000 customers over any 6-month period through the TV Network’s promotion of BioHarvest Israel, the TV Network will receive US$100,000, in Common Shares; or
·30,000 customers over any 6-month period through the TV Network’s promotion of BioHarvest Israel, the TV Network will receive US$200,000, in Common Shares,
such Common Shares to be issued at a price per Common Share equal to the closing price of the Common Shares immediately prior to such issuance.
MZ Group Agreement
Pursuant to the investor relations agreement dated February 15, 2024 (the “MZ Group Agreement”) between the Company and MZHCI, LLC (the “MZ Group”), the MZ Group provides investor relations services to the Company. In consideration for the MZ Group’s services:
·the MZ Group receives a monthly cash fee, in respect of which US$245,331 has been paid as at the date of this Annual Information Form;
·certain individuals providing the services from the MZ Group received an aggregate of 13,600 Options, with each Option exercisable at C$7.875 to acquire one (1) Common Share; and
45
·the MZ Group is entitled to receive performance-based cash bonuses, in respect of which US$30,000 has been paid as at the date of this Annual Information Form.
The MZ Group Agreement will have an initial term of three (3) months, subject to automatic renewal for an additional three (3) months, unless terminated by the Company or the MZ Group. If the MZ Group Agreement is not terminated after six (6) months, the MZ Group Agreement will automatically renew for an additional six (6) month term and continue to automatically renew every six (6) months, unless terminated by the Company or the MZ Group. The MZ Group Agreement may be terminated by the Company or the MZ Group providing thirty (30) days’ written notice to the other party prior to the end of the then-current term.
NAMES AND INTERESTS OF EXPERTS
Ziv Haft, Certified Public Accountants (Isr), a BDO Member Firm, provided an auditor’s report in respect to the Company’s audited annual consolidated financial statements for the financial year ended December 31, 2024. Ziv Haft, Certified Public Accountants (Isr) has advised that they are independent with respect to the Company within the meaning of Chartered Professional Accountants of British Columbia (CPABC) Code of Professional Conduct and the rules of the SEC and the Public Company Accounting Oversight Board (PCAOB) on auditor independence.
ADDITIONAL INFORMATION
Additional information, including directors’ and officers’ remuneration and indebtedness, principal holders of the Company’s securities and securities authorized for issuance under equity compensation plans, as applicable, are contained in the Company’s management information circular dated April 23, 2024 in respect of the Company’s annual general and special meeting of shareholders held on May 27, 2024.
Additional financial information is provided in the Company’s audited annual consolidated financial statements and management’s discussion and analysis for the financial year ended December 31, 2024, which are available under the Company’s profile on the System for Electronic Document Analysis and Retrieval + (“SEDAR+”) at www.sedarplus.ca.
Copies of all materials incorporated by reference herein and additional information relating to the Company are available under the Company’s profile on SEDAR+ at www.sedarplus.ca.
46
SCHEDULE “A”
AUDIT COMMITTEE CHARTER
(See attached)
BIOHARVEST SCIENCES INC.
AUDIT COMMITTEE CHARTER
(Amended and Restated as of September 20, 2024)
1.PURPOSE.
1.1The purpose of the Audit Committee (the “Committee”) of the board of directors (the “Board”) of BioHarvest Sciences Inc. (the “Company”) is to assist the Board with oversight of the Company’s accounting and financial reporting processes and the audit of the Company’s financial statements.
1.2The primary role of the Committee is to oversee the Company’s financial reporting and disclosure process. To fulfill this obligation, the Committee relies on:
(a)the Company’s executive officers and their employee designees (“management”) for the preparation and accuracy of the Company’s financial statements;
(b)both management and the Company’s personnel responsible for establishing effective internal controls and procedures to ensure the Company’s compliance with accounting standards, financial reporting procedures and applicable laws and regulations; and
(c)the Company’s independent auditors for an unbiased, diligent audit or review, as applicable, of the Company’s financial statements and the effectiveness of the Company’s internal controls.
The members of the Committee are not employees of the Company and are not responsible for conducting the audit or performing other accounting procedures.
2.COMMITTEE MEMBERSHIP.
2.1The Committee shall consist of three (3) or more directors. Each member of the Committee shall be “independent” in accordance with the requirements of Rule 10A-3 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”) and the rules of The Nasdaq Stock Market, LLC. No member of the Committee can have participated in the preparation of the Company’s financial statements at any time during the past three (3) years.
2.2Each member of the Committee must be financially literate and able to read and understand fundamental financial statements, including the Company’s balance sheet, income statement and cash flow statement. At least one member of the Committee must have past employment experience in finance or accounting, requisite professional certification in accounting or other comparable experience or background that leads to financial sophistication. At least one (1) member of the Committee must be an “audit committee financial expert” as defined in Item 407(d)(5)(ii) of Regulation S-K. A person who satisfies this definition of audit committee financial expert will also be presumed to have financial sophistication.
2.3The members of the Committee shall be appointed by the Board and shall serve for such term or terms as the Board may determine or until earlier resignation, removal or death. The Board may remove any member from the Committee at any time with or without cause.
3.MEETINGS AND PROCEDURES.
3.1A majority of the members of the entire Committee shall constitute a quorum. The Committee shall act on the affirmative vote of a majority of members present at the meeting at which a quorum is present. The Committee may request any officer or employee of the Company or the Company’s outside counsel or independent auditor to attend a meeting of the Committee or to meet with any members of, or consultants to, the Committee.
3.2The Board shall designate a member of the Committee as the chairperson. The Committee shall meet at least four (4) times a year at such times and places as it deems necessary to fulfill its responsibilities. The Committee shall report to the Board on its discussions and actions, including any significant issues or concerns that arise at its meetings, and shall make recommendations to the Board as appropriate. The Committee is governed by the same rules regarding meetings (including meetings in person or by telephone or other similar communications equipment), action without meetings, notice, waiver of notice, and quorum and voting requirements as are applicable to the Board as provided for in the Company’s Articles, as amended and/or restated from time to time.
3.3Committee shall meet separately, and periodically, with management and representatives of the Company’s independent auditors, and shall invite such individuals to its meetings as it deems appropriate, to assist in carrying out its duties and responsibilities. However, the Committee shall meet regularly without such individuals present.
3.4The Committee shall review this Charter at least annually and recommend any proposed changes to the Board for approval.
4.POWERS AND RESPONSIBILITIES.
4.1The Committee shall have the following authority and responsibilities:
(a)to: (i) select and retain an independent registered public accounting firm to act as the Company’s independent auditors for the purpose of auditing the Company’s annual financial statements, books, records, accounts and internal controls over financial reporting; (ii) set the compensation of the Company’s independent auditors; (iii) oversee the work done by the Company’s independent auditors; and (iv) terminate the Company’s independent auditors, if necessary in the Committee’s determination;
(b)to select, retain, compensate, oversee and terminate, if necessary, any other registered public accounting firm engaged for the purpose of preparing or issuing an audit report or performing other audit, review or attest services for the Company;
(c)to (i) approve all audit engagement fees and terms (with the power to sign any engagement letter providing for the same on behalf of the Company) and (ii) pre-approve all audit and permitted non-audit and tax services that may be provided by the Company’s independent auditors or other registered public accounting firms, and establish policies and procedures for the Committee’s pre-approval of permitted services by the Company’s independent auditors or other registered public accounting firms on an on-going basis;
(d)at least annually, to obtain and review a report by the Company’s independent auditors that describes: (i) the accounting firm’s internal quality control procedures; (ii) any material issues raised by the most recent internal quality control review, peer review or Public Company Accounting Oversight Board (“PCAOB”) review or inspection of the firm or by any other inquiry or investigation by governmental or professional authorities in the past five years regarding one or more audits carried out by the firm and any steps taken to deal with any such issues; and (iii) all relationships between the firm and the Company; and to discuss with the independent auditors this report and any relationships or services that may impact the objectivity and independence of the auditors;
(e)at least annually, to evaluate the qualifications, performance and independence of the
A-2
Company’s independent auditors, including an evaluation of the lead audit partner; and to assure the regular rotation of the lead audit partner at the Company’s independent auditors and consider regular rotation of the accounting firm serving as the Company’s independent auditors;
(f)to review and discuss with the Company’s independent auditors: (i) the auditors’ responsibilities under generally accepted auditing standards and the responsibilities of management in the audit process; (ii) the overall audit strategy; (iii) the scope and timing of the annual audit; (iv) any significant risks identified during the auditors’ risk assessment procedures; and (v) when completed, the results, including significant findings, of the annual audit;
(g)to review and discuss with the Company’s independent auditors: (i) all critical accounting policies and practices to be used in the audit; (ii) all alternative treatments of financial information within generally accepted accounting principles (“GAAP”) that have been discussed with management, the ramifications of the use of such alternative treatments and the treatment preferred by the auditors; and (iii) other material written communications between the auditors and management;
(h)to review and discuss with the Company’s independent auditors and management: (i) any audit problems or difficulties, including difficulties encountered by the Company’s independent auditors during their audit work (such as restrictions on the scope of their activities or their access to information); (ii) any significant disagreements with management; and (iii) management’s response to these problems, difficulties or disagreements; and to resolve any disagreements between the Company’s auditors and management;
(i)to review with management and the Company’s independent auditors: (i) any major issues regarding accounting principles and financial statement presentation, including any significant changes in the Company’s selection or application of accounting principles; (ii) any significant financial reporting issues and judgments made in connection with the preparation of the Company’s financial statements, including the effects of alternative GAAP methods; and (iii) the effect of regulatory and accounting initiatives and off-balance sheet structures on the Company’s financial statements;
(j)to inform the Company’s independent auditors as requested as to the Committee’s understanding of the Company’s relationships and transactions with related parties that are significant to the Company; and to review and discuss with the Company’s independent auditors the auditors’ evaluation of the Company’s identification of, accounting for, and disclosure of its relationships and transactions with related parties, including any significant matters arising from the audit regarding the Company’s relationships and transactions with related parties;
(k)to review with management and the Company’s independent auditors: (i) the adequacy and effectiveness of the Company’s internal controls, including any significant deficiencies or material weaknesses in the design or operation of, and any material changes in, the Company’s internal controls; (ii) any special audit steps adopted in light of any material control deficiencies; (iii) any fraud involving management or other employees with a significant role in such internal controls; (iv) the independent auditors’ attestation (as required) of the report on internal controls and the required management certifications to be included in or attached as exhibits to the Company’s Annual Report on Form 20-F or quarterly report on Form 6-K, as applicable;
(l)to review and discuss with the Company’s independent auditors any other matters required to be discussed by applicable requirements of the PCAOB and the Securities and Exchange Commission (“SEC”);
(m)to review and discuss with the Company’s independent auditors and management the
A-3
Company’s annual audited financial statements (including the related notes), the form of audit opinion to be issued by the auditors on the financial statements and the disclosure under “Management’s Discussion and Analysis of Financial Condition and Results of Operations” to be included in the Company’s Annual Report on Form 20-F before such Form 20-F is filed, and recommend to the Board whether the audited financial statements should be included in the Company’s Form 20-F and whether the Form 6-K should be filed with the SEC;
(n)to produce the audit committee report required to be included in the Company’s annual or other proxy statements;
(o)to review and discuss with the Company’s independent auditors and management the Company’s quarterly financial statements and management’s discussion and analysis to be included in the Company’s Quarterly Report on Form 6-K before such Form 6-K is filed; and to review and discuss the Form 6-K for filing with the SEC;
(p)to recommend to the Board policies for the Company’s hiring of employees or former employees of the Company’s independent auditors;
(q)to establish and oversee Company procedures for the receipt, retention and treatment of complaints received about the Company regarding accounting, internal accounting controls or auditing matters, or instances of fraud or unlawful conduct, and for the confidential, anonymous submission by Company employees of concerns regarding such matters;
(r)to review and discuss with management the material risks faced by the Company and the policies, guidelines and processes by which management assesses and manages the Company’s risks, including the Company’s major financial risk exposures and the steps management has taken to monitor and control such exposures;
(s)to oversee the Company’s compliance with applicable laws and regulations and to review and oversee the Company’s policies, procedures and programs designed to promote and monitor such legal and regulatory compliance;
(t)to review with the Company’s legal counsel, legal and regulatory matters, including legal cases against or regulatory investigations of the Company that could have a significant impact on the Company’s financial statements;
(u)to review, approve and oversee any transaction between the Company and any related person (as defined in Item 404 of Regulation S-K promulgated by the SEC) and any other potential conflict of interest situations on an ongoing basis, in accordance with Company policies and procedures, and to develop policies and procedures for the Committee’s approval of related party transactions; and
(v)to implement and oversee the Company’s cybersecurity and information security policies, including the periodic review of the policies and managing potential cybersecurity incidents.
5.OUTSIDE ADVISORS.
5.1The Committee shall have the authority, in its sole discretion, to retain and obtain the advice and assistance of independent outside counsel and such other advisors as it deems necessary to fulfill its duties and responsibilities under this Charter. The Committee shall set the compensation, and oversee the work, of any outside counsel and other advisors.
5.2The Committee shall receive appropriate funding from the Company, as determined by the Committee in its capacity as a committee of the Board, for the payment of compensation to the Company’s independent auditors, any other accounting firm engaged to perform services for the Company, any outside counsel and any other advisors to the Committee.
A-4
6.DELEGATION OF AUTHORITY.
6.1The Committee shall have the authority to delegate any of its responsibilities, along with the authority to take action in relation to such responsibilities, to one or more subcommittees as the Committee may deem appropriate in its sole discretion.
7.PERFORMANCE EVALUATION.
7.1The Committee shall conduct or otherwise participate in/respond to an annual evaluation of the performance of its duties under this Charter and shall present, or otherwise participate in, the results of the evaluation to the Board. The Committee shall conduct this evaluation in such manner as it deems appropriate.
8.CLAWBACK REQUIREMENTS.
8.1To the extent that the Company continues to be listed on an exchange on which securities are traded and subject to Rule 10D-1 of the Exchange Act, the Committee shall assist and advise the Board and the Compensation Committee thereof in enforcing the Company’s Executive Compensation Clawback Policy and related laws, rules and regulations.
9.DISCLOSURE OF CHARTER.
9.1This Charter and any amendments or restatements to this Charter will be made available on the Company’s website.
Adopted by the Board on September 20, 2024.
A-5

